Pleasecitethisarticleinpressas:S.Lamnini,etal.,Theroleoftheattritionmillingonthegrainsizeanddistributionofthecarbonnanotubes w w w . e l s e v i e r . e s / b s e c v
The role of the attrition milling on the grain size and distribution of the carbon nanotubes
in YSZ powders
Soukaina Lamnini
a,b, Zsolt Fogarassy
b, Zsolt Endre Horváth
b, Sára Tóth
c, Katalin Balázsi
b,∗, Csaba Balázsi
baDoctoralSchoolofMaterialScienceandTechnologies,ÓbudaUniversity,Bécsistr.96/B,1034Budapest,Hungary
bInstituteforTechnicalPhysicsandMaterialsScience,CentreforEnergyResearch,HungarianAcademyofSciences,Konkoly–ThegeM.
str.29-33,1121Budapest,Hungary
cWignerResearchCentreforPhysics,HungarianAcademyofSciences,Konkoly–ThegeM.str.29-33,1121Budapest,Hungary
a r t i c l e i n f o
Articlehistory:
Received14May2018 Accepted17October2018 Availableonlinexxx
Keywords:
MWCNT Milling
Yttria-stabilizedzirconia
a bs t r a c t
Theaimofpresentworkwastheexaminationoftheroleofthehighefficientattrition millingonthestructureandgrainsizeofZrO2–8mol.%Y2O3(YSZ)andonthedistribution ofmultiwallcarbonnanotubes(MWCNTs)inthesepowdermixtures.Themicrostructureof YSZ/MWCNTspowdermixtureswith1,5,10wt.%MWCNTswasinvestigated.Detailedstudy confirmedtheYSZgrainsizedecreaseandsimultaneouslythenon-destructivehomoge- nizationofMWCNTsintheceramicpowdermixtures.Thebesthomogenizationdegreewas achievedinthecaseofYSZ/1wt.%MWCNTpowder.Ramanscatteringmeasurementswere performedtoinvestigatetheatomicbondingandstructuralintegrityofcarbonnanotubes.
ThepresenceoftheGandDbandsineachsamplesat∼1590and∼1355cm−1confirmedthe unaffectedstructuralintegrityofMWCNTsafterthemillingprocess.InadditiontoRaman measurementsthehighresolutiontransmissionelectronmicroscopy(HRTEM)studieshave shownthatthestructureofMWCNTsremainsintactaftermilling.
©2018SECV.PublishedbyElsevierEspa ˜na,S.L.U.Thisisanopenaccessarticleunderthe CCBY-NC-NDlicense(http://creativecommons.org/licenses/by-nc-nd/4.0/).
Elroldelamoliendaporatricióneneltama ˜nodegranoyladistribución denanotubosdecarbonoenlospolvosYSZ
Palabrasclave:
MWCNT Fresado
Zirconiaestabilizadaconitria
r e su m e n
El propósitodeeste trabajo fuela evaluacióndelroldelamolienda poratriciónenla estructuraytama ˜nodegranodeZrO2–8mol.%Y2O3(YSZ)yladistribucióndetubosdecar- bonomultipared(MWCNTs)enlasmencionadasmesclasmixtaspulverizadas.Seinvestigó lamicroestructuradelospolvosYSZ/MWCNT.Elestudiodetalladoconfirmóladisminu- cióndeltama ˜nodegranoYSZy,simultáneamente,lahomogeneizaciónnodestructivade
∗ Correspondingauthor.
E-mailaddress:balazsi.katalin@energia.mta.hu(K.Balázsi).
https://doi.org/10.1016/j.bsecv.2018.10.001
0366-3175/©2018SECV.PublishedbyElsevier Espa ˜na,S.L.U.Thisisanopen accessarticleundertheCCBY-NC-NDlicense (http://
creativecommons.org/licenses/by-nc-nd/4.0/).
Pleasecitethisarticleinpressas:S.Lamnini,etal.,Theroleoftheattritionmillingonthegrainsizeanddistributionofthecarbonnanotubes MWCNTenlasmezclascerámicaspulverizadas.Elmejorgradodehomogenizaciónfue alcanzadoenelcasodeYSZal1%enpesodeMWCNT.Serealizaronmedicionesdedisper- siónRamanparainvestigarlauniónatómicaylaintegridadestructuraldelosnanotubos decarbono.LapresenciadelasbandasGyDencadamuestraa∼1590cm-1y∼1355cm-1, confirmóquelaintegridadestructuraldelosMWCNT,nofueafectadadespuésdelproceso demolienda.AdemásdelasmedicionesdeRaman,losestudiosdemicroscopíaelectrónica detransmisióndealtaresolución(HRTEM)demostraronquelaestructuradelosMWCNT permaneceintactadespuésdelamolienda.
©2018SECV.PublicadoporElsevierEspa ˜na,S.L.U.Esteesunart´ıculoOpenAccessbajo lalicenciaCCBY-NC-ND(http://creativecommons.org/licenses/by-nc-nd/4.0/).
Introduction
Investigationofadvancedceramics,namelyzirconiaandtheir compositesbecameanimportanttaskfortremendousscien- tificresearchdealingwithenergyproduction,conversionand storage.Theincorporationofanadditivephaseintozirconia matrixappearsasaverypromisingapproach,especiallyfor enhancingthethermo-mechanicalandtheelectricalproper- tiesofthecomposites[1].Therefore,advancedceramicsare widelyexploredforenergyapplicationsanddifferentceramic componentsareappliedtosolidoxidefuelcells,supercapac- itors,hydrogenstoragesystemsandphotovoltaicsolarcells [2]. Fuel cells are classified according totwo different cat- egories. The first one depends on the nature of the fuels usedforpowergeneration[3]suchashydrogen[4],methanol [5],fossilfuels[4]orbiomass-derivedmaterials[6].Thesec- ond category includes the composition of the catalysts to speeduptheelectrochemicalprocessesregardingthediffer- ent possibleworking temperature ofthe cell [7]. Thesolid oxidefuelcells (SOFC)operating athightemperature from 800◦Cto1000◦Careoneofthebestefficientpowergeneration devices devoted toproduce clean energywithout environ- mentaldamages [8].Yttria-stabilized zirconia(YSZ)isused tobethe most frequentlyelectrolytematerial forSOFC[9]
toconvertthechemicalenergyofthereactantsdirectlyinto electricity and heat. The output of the reaction is water.
Thehigh temperature can cause many problemsin terms ofanode-electrolyte–cathodedegradationandlifetimeofthe cells [10]. The main challenge is the decrease of the high workingtemperature of fuelcells. Liuet al.identified and analyzedthemainissuesresponsibleforcathodedegradation inSOFC[11].Themicrostructuralchangesattheinterfaceof LSM((LaxSr1−x)yMnO3)/YSZcathodeandYSZelectrolyteintro- ducedareductionoftheLSMcratersandtheformationofnew phasesoftheinsulatingzirconate.Thelackofoxygencon- tentintheairintroducedtothecathodegaswasattributed asamajorfactor responsibleofLSMcraters reduction.Ni- YSZcermetanodesarethemostcommonelectrodeforSOFC operatingathightemperature.ThemetallicNiparticlesare responsibleofelectronconductionproducedfromhydrogen oxidationaccordingtothefollowingreaction[12]:
2H2+2O2−→2H2O+4e− (1)
ThehighporositydensityisrecommendedforNi-YSZcer- metanodestoensuretherequiredperformanceofthecells.
Indeed,TriplePhase Boundary(TPB)isthepoint whereNi, YSZ, and the poremeet and participateinelectrochemical reaction[13].Theproducedwatermoleculeistransferredout- sideoftheanodethroughthepores.Ni-YSZanodesworkas acatalystusedtoreformthemethane.Therefore,impurities suchascarbondepositionnamedalso“cooking”or“sulphur deposition”remainthemajorissuesresponsibleforthefast anodedeterioration.Thepresenceofsulphurandintensive sulphurdepositioncontributedtothedelaminationofanode layerandblockthenickelgrains.Thisfactcausedthelimi- tationofthehydrogenatom’smovementandthusleadstoa significantdecreaseofthecellefficiency.Chengetal.found thattheformationofsulphide(Ni3S2,NiS)canbelimitedbya highcoolingrateabout70◦C/min[14].Whilethecarbondepo- sitionontheporousanodecanoccurindifferentformssuch as fibres,whiskers orgraphitic carbon causing micro mor- phologicalchangesoftheanoderesultingindeactivationor breakdownofthecatalysts[15].Manyrecentstudiesproved thatthetemperatureofsinteringisanimportantparameter inobtainingasmallgrainedstructureforabetterperformance ofSOFCelectrolyte[16].
Oneoftheadvancedapplicationofceramicsassolarcells resulted inlowthermalemittance and increasedcorrosion resistanceatelevatedtemperaturesbyapplyingoftheaddi- tional layersofzirconium thinfilm between substrate and absorber[17].Supercapacitorsaredevelopedonthebasisof zirconiadopedCNTorgraphenecharacterizedbyenhanced mechanical andelectrochemicalproperties. Amiranteetal.
concluded that the capacitance of supercapacitor materi- als isdepending strongly on the carbon nanotubes (CNTs) orientation [18]. Alves et al. studied the new concept ofa ternarycompositeconsistingofZrO2nanoparticles,reduced grapheneoxide(rGO) andinsitupolymerizedPyrrole (PPy).
It was proved that the addition of zirconiainfluenced the morphology and increased the porosity of the electrodes resultinginremarkableenhancementinthecapacitanceof the supercapacitor [19]. On the other hand, a significant improvementin bothcompressive and torsionalresistance hasbeenreported.Differentalloysofmetalhydridesmate- rials have been studied in the literature namely: cerium, lanthanum and nickel (Ce-La-Ni) or magnesium hydride combined with zirconium oxide and single-walled carbon nanotubes. However, this modern method of storage has somelimitationsashighthermalstability,difficultytodecom- pose the hydride system into metal and hydrogen gas or apoorkineticsrateofhydrogenationanddehydrogenation.
El-Eskandaranyetaletal.studiedthedopingeffectofMgH2
Pleasecitethisarticleinpressas:S.Lamnini,etal.,Theroleoftheattritionmillingonthegrainsizeanddistributionofthecarbonnanotubes nano-powderswith10wt.% ofnanocrystalliteZrNi5 onthe
hydrogenation/dehydrogenationbehaviour[20].Theseresults showed improved properties with a short absorption time of1 and 10min torelease 5.3wt.% ofH2. This study con- firmedthatthezirconiumtetrachloride(ZrCl4)asacatalyst intoMgH2exhibitedanotablereductioninthedehydrogena- tion and rehydrogenation temperature. More importantly, ZrCl4 prevented thegrain growth of the hydridefor better cyclicperformance[21].
The aim of our study was the development of YSZ/MWCNTs as nickel free SOFC material. This work presents the mainresults offirst preparationstep; milling andhomogenization.Thedetailedstructuraldesignofmilled YSZ/MWCNT powder mixtures with various addition of MWCNTsfrom 1to10wt.% was performed. Thestructural andmorphologicalpropertiesofpowdermixtureswereinves- tigatedbyscanningelectronmicroscopy(SEM),transmission electron microscopy (TEM), high resolution transmission electron microscopy (HRTEM), X-ray diffraction (XRD) and Ramanspectroscopy.
Experimental
Preparationofpowdermixtures
ZrO2 –8mol.%Y2O3 (yttria-stabilizedzirconia (YSZ),Sulzer MetcoAMDRY6643) with anaveragegrain size40mwas usedasbasematerialforpowdermixturepreparation(Table1, Fig.1a).Commerciallyavailablemultiwallcarbonnanotubes (MWCNT,typeNC3100TM,Nanocyl)withanaveragediameter 9.5nmandanaveragelength1.5mwereaddedtozirconia
Table1–Compositionofreferencesample andYSZ/MWCNTspowders.
Sample YSZ(wt.%) MWCNT(wt.%)
YSZ-100 100 0
YSZ-99 99 1
YSZ-95 95 5
YSZ-90 90 10
powderatdifferentconcentrations(1,5and10wt.%)(Table1, Fig.1b,c).Highefficiencyattritormilling(UnionProcess,type 01-HD/HDDM) wasused(4000rpm, 5h)forpowdermixture preparation.Eachbatchwith80gfinalproductwasmixedin zirconiatank(750cm3)by130gethanoland280mLzirconia balls(eachof1mmdiameter).Theobtainedpowdermixtures were driedat172◦C for25minutes, then sievedby100m mesh(Fig.1d).
Characterizationmethods
Thephase analysiswas performedbyX-raydiffractometry (XRD)withCuK␣radiationusingaBrukerAXSD8Discover diffractometerequippedwithGöbel-mirrorandascintillation detector.Energydispersive X-rayspectroscopy (EDS,Rontec Si(Li)detectorwithultrathinwindow)wasusedforelemen- talidentificationofthepowdermixturesafter5hofmilling.
The morphology and the characterization of the powders werestudiedbyScanningElectronMicroscopy(SEM,LEO1540 XB)andTransmissionElectronMicroscopy(TEM,PhilipsCM- 20) and HighResolution Transmission Electron Microscopy (HRTEM, JEOL 3010) operating at 200 and 300kV, respec- tively.Ramanspectroscopyisawell-known,non-destructive
40 μm
2 μm 20 nm
400 nm
a
c d
b
Fig.1–Morphologyofthestartingmaterialsandpowders.(a)SEMimageofYSZasreceived(SE,MAG500×HV5kV), (b)SEMimageofMWCNTasreceived(SE,MAG10000×HV5kV),(c)TEMimageofMWCNTasreceived(at200kV) and(d)SEMimagemilledYSZafter5h(SE,MAG5000×HV5kV).
Pleasecitethisarticleinpressas:S.Lamnini,etal.,Theroleoftheattritionmillingonthegrainsizeanddistributionofthecarbonnanotubes
20 40 60
(311)
(220)
(200)
(111)(111)(-111)
80 2θ
Intensity ua.
YSZ - 90
monoclinic zirconia (JCP2:00-037-1484) cubic zirconia (JCP2:00-049-1642) Si
CNT
YSZ - 95
YSZ - 99
YSZ - 100 (reference)
Fig.2–XRDpatternsofYSZreferenceandYSZ/MWCNTpowders.
methodtoinvestigatetheatomicbondingpropertiesandthe microstructure.Renishaw1000Bmicro-Ramanspectrometer attachedtoaLeicaDM/LMmicroscopewasusedtoexamine Ramanspectroscopyatroomtemperatureinthewavenum- berrangeof150–3500cm−1with488nmlaserexcitation.The spectralresolutionofthesystemis2.5cm−1andthediameter oftheexcitationspotis1m.
Results and discussion
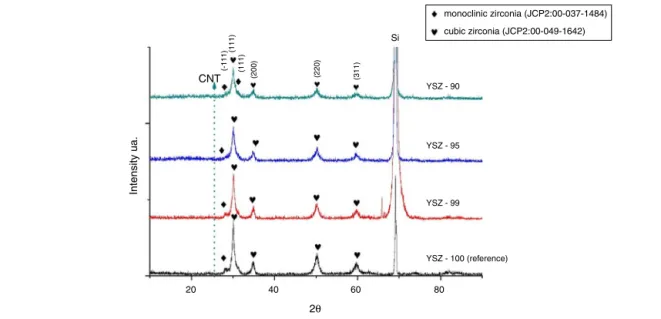
TheYSZbasedpowdermixtureswithvariousMWCNTaddi- tives(1,5and10wt.%)werepreparedbyattritormilling(Fig.1 andTable1).Theresultofthephaseexaminationoftheref- erenceYSZandmilledpowdermixturesisshowninFig.2.
Themilledpowderswereconsistedmainlyofcubiczirconia (JCP2:00-049-1642) and minor monoclinic zirconia (JCP2:00- 037-1484)phases(Fig.2).Thecubiczirconiaisindicatedby the mainlines appearedat2Â of30.24◦, 34.88◦, 50.17◦ and 59.69◦.While,theminormonoclinicphasewasobservedat 2Â of28.36◦ and 31.12◦.As forfirst observation, the milling processdidnotresultedinphasetransformationonlygrain decreaseoccured.YSZreferencebasematerialexhibitedwell dispersedparticleswithsharpandirregularshape(Fig.1).The morphologicalstudyofMWCNTdemonstratedtheiragglom- eratedbundlestructure.Ontheother hand,theadditionof MWCNTstozirconiamatrixintroducedasignificantchange inthesurfacetopologyandthedistribution ofthe zirconia grains,leadingtovariousformsandshapescomparedtothe basematerial(Fig.3).
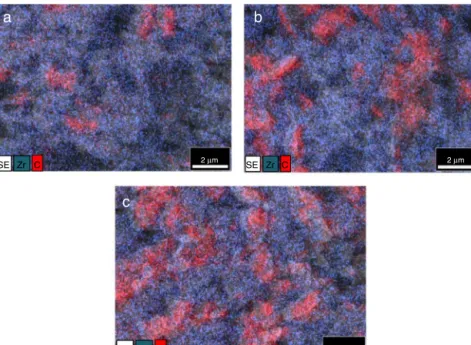
The EDS elemental analysis illustrated the features of MWCNTsdistributioninzirconiamatrix (Fig. 4).Thisstudy confirmed that the MWCNTs formed agglomerations with increasingtendencywithMWCNTsaddition increase(YSZ- 99→YSZ-95→YSZ-90).TheMWCNTsagglomerations(C-red, Fig. 4) were surrounded by ultrafine rounded zirconium
(Zr-blue).Structuralinvestigationsofzirconiapowderbefore andaftermillingprocesswerecarriedoutbyTEM(Fig.5).The TEMinvestigationdemonstratedthedecreaseofYSZparticle sizeinaverage50mto400nm.
StructuralobservationsofthemixedpowdersYSZ-99,YSZ- 95, YSZ-90are shown inFig. 6. Thisresultshows that the small zirconia grains(<100nm)and particles (>200nm) are embeddedintoMWCNTsagglomerationsandnetworks.The agglomeration,thedamageorthedefectofMWCNTshasbeen reportedasabigissueresponsibleofasignificantlackinthe reinforcementofceramiccompositeswithMWCNT.Fromthe pointofMWCNTsstructurethenon-destructivemillingpro- cesswasconfirmedbyHRTEMstudy.TheMWCNTsappeared asafinefibresformingseveralagglomerationswithanapprox- imatelengthof2.5m.Eachfibreconsistedofanaverageinner diameter 3.8nm, anaverage outer diameter9.13nmand a numberoflayersvariatedfrom7to9(Fig.6b).Theseresultsare inaccordancewiththestudyperformedbyMelkaetal.[22].
Theyconfirmedtherefinementofthegrainsizesupto2wt.%
ofMWCNTusedforpowdermixtureprocessing[22].Indeed, whentheagglomerationofMWCNToccurs,therealamount of MWCNTslocatedin the grain boundariesisdecreasing.
Therefore, the agglomerated CNTsintroduce heterogeneity anddecreasetheefficiencyofthecomposite[23–25].
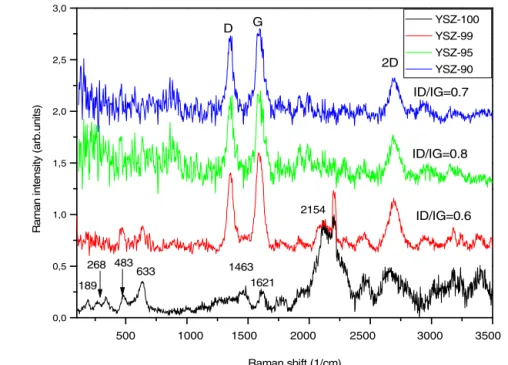
Scanning Raman spectroscopy measurementswere per- formed toconfirmthe presence ofMWCNTinthe powder mixtures and determine structural changes caused by the millingprocess.FourtypicalRamanspectraareshowninthe 150–3500cm−1wavenumberregion,whichweremeasuredon powdermixtureswithdifferentMWCNTcontent(Fig.7).In caseofpure YSZ(reference, YSZ-100,blackline) scattering peaksat189,268,341,380,483,633cm−1relatedtoyttriasta- bilizedzirconiawhichareinaccordancewithliteratureworks [26,27].CharacteristicRamanpeaksofYSZarelessintensive or completely absent in case of YSZ/MWCNTs powders (YSZ-99,YSZ-95,YSZ-90)inthe100to700cm−1wavenumber
Pleasecitethisarticleinpressas:S.Lamnini,etal.,Theroleoftheattritionmillingonthegrainsizeanddistributionofthecarbonnanotubes 400 nm
400 nm
400 nm
a b
c
Fig.3–SEMimagesofmilledpowders.(a)YSZ/1wt.%MWCNT,(b)YSZ/5wt.%MWCNTandc)YSZ/10wt.%MWCNT (SE,MAG5000×HV5kV).
a
SE SE
SE
Zr Zr
Zr
C C
C
c
2 μm
2 μm
2 μm
b
Fig.4–EDSmappingshowingthedispersionofMWCNTsinYSZ.(a)YSZ/1wt.%MWCNT,(b)YSZ/5wt.%MWCNT and(c)YSZ/10wt.%MWCNT.Colours:C-redandZr-blue.
region.Additionalstrongpeakshavebeen observedaround 1590cm−1(Gband)and1355cm−1(Dband),whicharerelated totheMWCNT[28].TheGbandisrelatedtothestretching vibrationofthesp2C–Cbonds,whiletheDbandisassociated withthepresenceofstructuraldefectsintheMWCNT.Raman spectraalsoexhibitedawidebandathigherwavenumbers,
the so called 2D band, which appears at 2687, 2684 and 2690cm−1for1,5,10wt.%MCWNTcontent,respectively.The 2D band feature originatesin a two-phonon,second-order Raman scattering process in carbon nanotubes [29]. The intensityratioofDandGband(ID/IG)hasbeenwidelyused to investigate the structural purity (structural defects and
Pleasecitethisarticleinpressas:S.Lamnini,etal.,Theroleoftheattritionmillingonthegrainsizeanddistributionofthecarbonnanotubes
200
a b
220 200
220 111
111
200 nm 200 nm
Fig.5–TEMimagesofYSZwithSAEDdiffractionininsert.(a)Referencebeforemillingand(b)referenceaftermilling.The SAEDconfirmedthecubiczirconia(111,220,200)inbothcases.
MWCNTs
c
a b
Zirconia particles Zirconia grains
200 nm
200 nm 50 nm
5 nm
Fig.6–TEMimagesofYSZ/MWCNTs.(a)YSZ/1wt.%MWCNT,(b)YSZ/5wt.%MWCNTwithdetailofMWCNTininsertand (c)YSZ/10wt.%MWCNT.
disordering)ofMWCNTsor thepresenceofagglomerations [30,31], hence the intensityofD bandis defectdependent.
In principle, the larger the number of defects, the higher theDbandintensity.Peakpositionsandintensityofdistinct scattering bands were determined by Lorentzian fitting of RamanspectrashowninFig.7.Althoughsuchprocedurehas someuncertaintybecauseofthenumberoffreeparameters,
itcanprovideadditionalinformationabouttheYSZ/MWCNTs powders.WefoundthattheID/IG ratiois0.6forYSZ-99,0.8 forYSZ-95and0.7forYSZ-90,whichvaluesareingoodaccor- dance with previous morphological observations showing the presence of structural defects and agglomerations in the YSZ/MWCNTspowders asaresultofintensivemilling.
Amongthepowdermixtures,YSZ-95containsmorestructural
Pleasecitethisarticleinpressas:S.Lamnini,etal.,Theroleoftheattritionmillingonthegrainsizeanddistributionofthecarbonnanotubes 0,0
0,5 1,0 1,5 2,0 2,5 3,0
ID/IG=0.7
ID/IG=0.8 2D
YSZ-100 YSZ-99 YSZ-95 YSZ-90
Raman intensity (arb.units)
Raman shift (1/cm) 189
268 483
633 1463
1621 2154
D G
ID/IG=0.6
500 1000 1500 2000 2500 3000 3500
Fig.7–Ramanspectrausingalaserexcitationwavelengthat488nmforreferenceandYSZ/MWCNTs.
defectscomparedtotheothersaccordingtoitshighestID/IG
ratio(0.8).
Conclusion
TheaimofourstudywasthedevelopmentofYSZ/MWCNTsas nickelfreeSOFCmaterial.Thisworkshowedthemainresults offirst preparation step; millingand homogenization. The influenceofMWCNTadditionandmillingprocessonstruc- tureanddesignofcompositeswerestudied.ZrO2–8mol.%
Y2O3(YSZ)powdersweremixedwith1,5and10wt.%MWC- NTsandmilledbyhighattritionmillingat4000rpminethanol for5h.Thestructuralandmorphologicalinvestigationsofas- receivedzirconiapowderconsistedoftheparticleswithsharp andirregular shape.In fact,the effectofmillingonthe as receivedzirconiaresultedinaconsiderabledecreaseforpar- ticlesizewithoutphasetransformationofYSZparticles.The phaseanalysisprovedtheexistenceoftwophases;cubiczirco- niaasamainphaseandasmallfractionofmonocliniczirconia asaminorphase.Theparticlesizeshowedarefinementfrom anaverageof50mto400nm.However,nosignificantdam- ageorstructuralchangesaffectedMWCNTsaftermillingwere observed.TheMWCNTswereobservedasafinefibresforming several agglomerations, networkswith approximate length 2.5m,eachfibreconsistedofanaveragediameter3.8nm,an averageouterdiameter9.13nm.Thenumberofcarbonlayers variedfrom7to9.Ramanspectroscopy resultsshowedthe presenceoftheGandDbandsforallpowdermixturesatpeak positionsof∼1590and∼1355cm−1respectively.Thesebands confirmedthestructuralintegrityofMWCNTafterthemilling process.
Thenovelresultsarefollows:
• millingprocessdidnotresultinphasetransformationof YSZonlygraindecrease;
• theadditionofMWCNTstozirconiamatrixintroducedasig- nificantchangeinthesurfacetopologyandthedistribution ofthezirconiagrains;
• from the point of MWCNTs structure, non-destructive millingprocesswasconfirmedbyHRTEMstudy;
• thestructuralintegrityofMWCNTsafterthemillingprocess wasconfirmedbyRamanmeasurementstoo.
Acknowledgement
Ms. Soukaina Lamnini thanksfor Stipendium Hungaricum scholarship.TheauthorsacknowledgethesupportfromCER- ANEA(FLAG-ERA)project.Furthermore,theauthorswouldlike tothankMr.LeventeIllésforSEMandEDSmeasurements.The authorsarealsogratefultoMr.ViktorVargaforhishelpwith samplepreparation.
references
[1]N.P.Padture,Multifunctionalcompositesofceramicsand single-walledcarbonnanotubes,Adv.Mater.21(2009) 1767–1770.
[2]O.Tatsuki,S.Mrityunjay,EngineeredCeramics:Current StatusandFutureProspects,292,1sted.,JohnWilleyand Sons,Hoboken,NJ,2016,pp.291–331,978-1-119-10040-9.
[3]C.S.Spiegel,Designing&buildingfuelcells,42,1sted., McGraw-HillEducation:Twopennplaza,NewYork,2007,pp.
35–51,978-0071489775.
[4]E.D.Wachsman,K.T.Lee,Loweringthetemperatureofsolid oxidefuelcells,Science334(2011)935–939.
[5]N.Laosiripojana,S.Assabumrungrat,Catalyticsteam reformingofmethane,methanol,andethanoloverNi/YSZ:
thepossibleuseofthesefuelsininternalreformingSOFC,J.
PowerSources163(2007)943–951.
Pleasecitethisarticleinpressas:S.Lamnini,etal.,Theroleoftheattritionmillingonthegrainsizeanddistributionofthecarbonnanotubes [6] T.A.KamelHalouani,Analyticalmodelingofpolarizationsin
asolidoxidefuelcellusingbiomasssyngasproductasfuel, Appl.Therm.Eng.27(2007)731–737.
[7] A.Alaswad,A.Palumbo,M.Dassisti,A.G.Olabi,FuelCell Technologies,Applications,andStateoftheArt.AReference Guide.ReferenceModuleinMaterialsScienceandMaterials Engineering,2016,http://dx.doi.org/10.1016/B978-0-12- 803581-8.04009-1.
[8] G.Cacciola,V.Antonucci,S.Freni,Technologyupdateand newstrategiesonfuelcells,J.PowerSources100(2001)67–79.
[9] L.Mingfei,D.Dehua,P.Ranran,G.Jianfeng,D.Juan,L.
Xingqin,M.Guangyao,YSZ-basedSOFCwithmodified electrode/electrolyteinterfacesforoperatingattemperature lowerthan650◦C,J.PowerSources180(2008)215–220.
[10]O.Tatsuki,S.Mrityunjay,EngineeredCeramics:Current statusandFutureProspects,422,1sted.,JohnWilleyand Sons,Hoboken,NJ,2016,pp.415–425,978-1–119-10040-9.
[11]Y.L.Lium,A.Hagen,R.Barfod,M.Chen,H.J.Wang,F.W.
Poulsen,P.V.Hendriksen,Microstructuralstudieson degradationofinterfacebetweenLSM–YSZcathodeandYSZ electrolyteinSOFCs,SolidStateIon180(2009)
1298–1304.
[12]T.Priyatham,B.Ranjit,Synthesisandcharacterizationof nanocrystallineNi–YSZcermetanodeforSOFC,Mater.
Charact.61(2010)54–58.
[13]S.P.Jiang,Y.Y.Duan,J.G.Love,Fabricationof
high-performanceNi/Y2O3–ZrO2cermetanodesofsolid oxidefuelcellsbyionimpregnation,J.Electrochem.Soc.149 (2002)1175–1186.
[14]Z.Cheng,M.Liu,Characterizationofsulfurpoisoningof Ni–YSZanodesforsolidoxidefuelcellsusinginsituRaman microspectroscopy,SolidStateIon178(2007)925–935.
[15]K.M.Shirjeel,B.L.Seung,H.S.Rak,W.L.Jong,T.L.Hyosung, S.J.Parka,Fundamentalmechanismsinvolvedinthe degradationofnickel–yttriastabilizedzirconia(Ni–YSZ) anodeduringsolidoxidefuelcellsoperation:areview, Ceram.Int.42(2016)35–48.
[16]M.Tanhaei,M.Mozammel,Yttria-stabilizedzirconiathin filmelectrolytedepositedbyEB-PVDonporousanode supportforSOFCapplications,Ceram.Int.43(3)(2017) 3035–3042.
[17]B.Usmani,V.Vijay,R.Chhibber,A.Dixit,Optimizationof sputteredzirconiumthinfilmsasaninfraredreflectorfor useinspectrally-selectivesolarabsorbers,ThinSolidFilms 627(2017)17–25.
[18]R.Amirante,E.Cassone,E.Distaso,P.Tamburrano,Overview onrecentdevelopmentsinenergystorage:mechanical, electrochemicalandhydrogentechnologies,EnergyConver.
Manag.132(2017)372–387.
[19]A.P.P.Alves,R.Koizumi,A.Samanta,L.D.Machado,A.Singh, D.S.Galvao,G.G.Silva,Ch.S.Tiwaryand,P.M.Ajayan, One-stepelectrodeposited3D-ternarycompositeofzirconia nanoparticlesrGOandpolypyrrolewithenhanced
supercapacitorperformance,NanoEnergy31(2017)225–232.
[20]S.M.El-Eskandarany,E.Shaban,H.Al-Matrouk,M.
Behbehani,A.Alkandary,F.Aldakheel,A.Naser,S.A.Ahmed, Structure,morphologyandhydrogenstoragekineticsof nanocompositeMgH2/10wt%ZrNi5powders,Mater.Today Energy3(2017)60–71.
[21]S.Kumar,etal.,SurfacemodificationofMgH2byZrCl4to tailorthereversiblehydrogenstorageperformance,Int.J.
HydrogenEnergy(2017),http://dx.doi.org/10.1016/j.
ijhydene.2017.01.193.
[22]L.Melk,etal.,Nanoindentationandfracturetoughnessof nanostructuredzirconia/multi-walledcarbonnanotube composites,Ceram.Int.(2014),http://dx.doi.org/10.1016/j.
ceramint.2014.10.060.
[23]A.Gallardo-Lopez,A.Morales-Rodríguez,J.Vega-Padillo,R.
Poyatob,A.Munoz,A.Domínguez-Rodrígueza,Enhanced carbonnanotubedispersionin3YTZP/SWNTscomposites anditseffectonroomtemperaturemechanicaland electricalproperties,J.AlloysCompd.682(2016)70–79.
[24]A.Duszová,J.Dusza,K.Tomsek,J.Morgiel,B.Gurdial,J.
Kuebler,Zirconia/carbonnanofibercomposite,Scr.Mater.58 (2008)520–523.
[25]A.Duszová,J.Dusza,K.Tomsek,J.Morgiel,B.Gurdial,J.
Kuebler,Microstructureandpropertiesofcarbon nanotube/zirconiacomposite,J.Eur.Ceram.Soc.28(2008) 1023–1102.
[26]A.Ghosh,A.K.Suri,M.Pandey,S.Thomas,T.R.RamaMohan, B.T.Rao,Nanocrystallinezirconia-yttriasystem–aRaman study,Mater.Lett.60(2006)1170–1173.
[27]J.Cai,Y.S.Raptis,E.Anastassakis,Stabilizedcubiczirconia:a Ramanstudyunderuniaxialstress,Appl.Phys.Lett.62 (1993)2781.
[28]L.Bokobza,J.Zhan,Ramanspectroscopiccharacterizationof multiwallcarbonnanotubesandofcomposites.eXPRESS Poly,Letters6(2012)601–608.
[29]M.S.Dresselhaus,etal.,Ramanspectroscopyofcarbon nanotubes,Phys.Rep.409(2005)47–99.
[30]E.Bódis,I.Cora,C.Balázsi,P.Németh,Z.Károly,S.Klébert,P.
Fazekasa,A.M.Keszlera,J.Szépvölgyia,Sparkplasma sinteringofgraphenereinforcedsiliconcarbideceramics, Ceram.Int.43(2017)9005–9011.
[31]E.F.Antunesa,A.O.Lobo,E.J.Corata,V.J.Trava-Airoldia,A.A.
Martin,C.Verısimo,Comparativestudyoffirst-and second-orderRamanspectraofMWCNTatvisibleand infraredlaserexcitation,Carbon44(2006)2202–2211.