PLANT PHYSIOLOGY
Az Agrármérnöki MSc szak tananyagfejlesztése TÁMOP-4.1.2-08/1/A-2009-0010
Principal Manuals in Plant Physiology
Arteca, R.N. (1996): Plant Growth Substances. Principles and Applications. Chapman & Hall, New York, p. 332.
Davies, P.J. (Ed.) (2004): Plant Hormones. Biosynthesis, Signal Transduction, Action! Kluwer Academic Publishers, Dordrecht/Boston/London, p. 750.
Hopkins, W.G., Hüner, N.P.A. (2009): Introduction to Plant Physiology. John Wiley & Sons, Inc., Hoboken, p. 503.
Salisbury, F.B., Ross, C.W. (1992): Plant Physiology.
Wadsworth Publishing Co., Belmont, p. 682.
Taiz, L., Zeiger, E. (2010): Plant Physiology. Sinaurer
Associates Inc., Publishers, Sunderland, p. 782 + 137.
Water and Plant Cells,
Water Potential
Overview
1. Water in Plant Life
2. The Structure and Properties of Water
3. Water Movement by Diffusion, Osmosis and Bulk Flow
4. Hydrostatic Pressure and Osmotic Pressure are two Components of Water Potential
5. Methods for Measurement of Water Potential
1. Water in Plant Life
1.1. Without water life could not exist
1.2. The actual water content of different plant cells, tissues and plant parts is variable
1.3. The productivity of various ecosystems depends on annual precipitation
1.4. Water loss to the atmosphere is an inevitable
consequence of photosynthesis
Source: Taiz L., Zeiger E. (2010): Plant Physiology. p. 68.
Productivity of various ecosystems as a function of annual precipitation
Source: Taiz L., Zeiger E. (2010): Plant Physiology. p. 97.
Water pathway through the leaf
2. The Structure and Properties of Water
2.1. Water has unique physical and chemical properties 2.2. The thermal properties of water are biologically important
2.3. Water is the universal solvent
2.4. Polarity of water molecules results in cohesion and
adhesion
Source: Hopkins W.G., Hüner N.P.A. (2009): Introduction to Plant Physiology. p. 2.
Some chemical properties of water compared with other molecules of
similar molecular size
Source: Hopkins W.G., Hüner N.P.A. (2009): Introduction to Plant Physiology. p. 2.
A) Structure of a water molecule
B) Hydrogen bonds among water molecules
Source: Hopkins W.G., Hüner N.P.A. (2009): Introduction to Plant Physiology. p. 4.
Solvent properties of water molecules
Source: Taiz L., Zeiger E. (2010): Plant Physiology. p. 70.
A) The shape of a droplet placed on a solid surface
B) Observation of capillarity
3. Water Movement by Diffusion, Osmosis and Bulk Flow
3.1. Diffusion is the net movement of molecules by random thermal agitation
3.2. Diffusion is effective over short distances 3.3. Osmosis is the diffusion of water across a selectively permeable membrane
3.4. Bulk flow is driven by hydrostatic pressure
Source: Hopkins W.G., Hüner N.P.A. (2009): Introduction to Plant Physiology. p. 6.
Diffusion is accomplished through the random thermal motion of
individual molecules
Source: Hopkins W.G., Hüner N.P.A. (2009): Introduction to Plant Physiology. p. 7.
Osmosis is the directed movement of the solvent molecules (usually
water) across a selectively permeable membrane
Source: Taiz L., Zeiger E. (2010): Plant Physiology. p. 71.
A seal syringe can be used to create positive and negative pressures in
fluids such as water
Source: Taiz L., Zeiger E. (2010): Plant Physiology. p. 71.
4. Hydrostatic Pressure and Osmotic Pressure are two Components of Water Potential
4.1. The hydrostatic pressure developed by osmosis can be demonstrated using an osmometer
4.2. The plant cell is a special osmotic system
Source: Salisbury F.B., Ross C.W. (1992):
Plant Physiology. p. 44.
a) A mechanical osmometer in a beaker
b) A cell as an osmotic system
Source: Taiz L., Zeiger E. (2010): Plant Physiology. p. 76.
The concept of water potential and its components
Source: Taiz L., Zeiger E. (2010): Plant Physiology. p. 76.
The concept of water potential and its components
Source: Taiz L., Zeiger E. (2010): Plant Physiology. p. 76.
The concept of water potential and its components
Source: Taiz L., Zeiger E. (2010): Plant Physiology. p. 76.
The concept of water potential and its components
5. Methods for Measurement of Water Potential
5.1. Water potential can be measured in any plant tissues and organs
5.2. Methods for measurement of water potential:
psychrometer, pressure chamber, cryoscopic osmometer,
and pressure probe
Source: Taiz L., Zeiger E. (2010): Plant Physiology. Web material, http://5e.plantphys.net
The use of isopiestic psychrometry to measure the water potential of a
plant tissue
Source: Taiz L., Zeiger E. (2010): Plant Physiology. Web material, http://5e.plantphys.net
The pressure chamber method for measuring plant water potential
Source: PMS Instrument Company, 1725 Geary Street SE, Albany, OR 97322 USA, http://pmsinstrument.com
The concept of a commercial water potential meter based on pressure
chamber method
Source: PMS Instrument Company, 1725 Geary Street SE, Albany, OR 97322 USA, http://pmsinstrument.com
'Pump-up chamber': the commercial water potential meter
Source: PMS Instrument Company, 1725 Geary Street SE, Albany, OR 97322 USA, http://pmsinstrument.com
Measuring of water potential using 'Pump-up chamber'
Source: Taiz L., Zeiger E. (2010): Plant Physiology. Web material, http://5e.plantphys.net
A cryoscopic osmometer measures the concentration of total dissolved
solutes by measuring the freezing-point depression of a solution
Source: Taiz L., Zeiger E. (2010): Plant Physiology. Web material, http://5e.plantphys.net
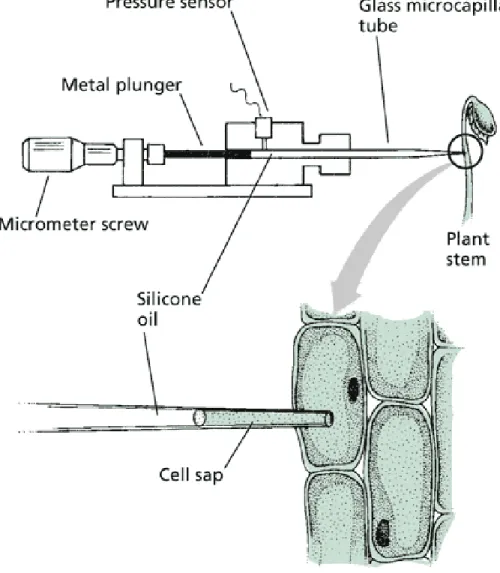
Use of the micromanometer, a pressure probe, to measure cell turgor
pressure
Source: Taiz L., Zeiger E. (2010): Plant Physiology. Web material, http://5e.plantphys.net