1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 Selected Papers from the 2nd Radiocarbon in the Environment Conference, Debrecen, Hungary, 3-7 July 2017
© 2018 by the Arizona Board ofRegents on behalf o f the University of Arizona
ISOTOPE ANALYTICAL CHARACTERIZATION OF CARBON-BASED NANOCOMPOSITES
T ib o r S zab ó • Ja n o v ic s R ó b e r t1 • M a ria n n a T ú r i1 • Is tv á n F u t ó 1 • Is tv á n P a p p 1 M ih á ly B r a u n • K ris z tiá n N é m e th 3 • G e rg ő P é te r S z e k e re d • A n ik ó K in k a 3 • A n n a S z a b ó 3 • K lá r a H e r n á d i • K a ta H a jd u 1,2 • L ász ló N a g y 2 • L ász ló R in y u 1
'isotope Climatology and Environmental Research Centre (ICER), Institute for Nuclear Research, Hungarian Academy of Sciences, Bem tér 18/c, Debrecen 4026, Hungary.
2Department of Medical Physics and Informatics, University of Szeged, Rerrich B. tér 1, Szeged 6720, Hungary.
d e p a rtm e n t of Applied and Environmental Chemistry, University of Szeged, Rerrich B. tér 1, Szeged 6720, Hungary.
ABSTRACT. Carbon-based nanomaterials of different dimensions (1-3D, tubes, bundles, films, papers and sponges, graphene sheets) have been created and their characteristic properties are discussed in the literature. Due to their unique advantageous, tunable properties these materials became promising candidates in new generations of applica
tions in many research laboratories and, recently, in industries as well. Protein-based bio-nanocomposites are referred to as materials of the future, which may serve as conceptual revolution in the development of integrated optical devices, e.g. optical switches, microimaging systems, sensors, telecommunication technologies or energy harvesting and biosensor applications. In our experiments, we designed various carbon-based nanomaterials either doped or not doped with nitrogen or sulfur during catalytic chemical vapor deposition synthesis. The amounts of heteroatoms in the composites were determined by radioanalytical and isotope analytical methods and their physico-/chemical prop
erties were compared. Based on 14C-analytics the yield of protein functionalization was also evaluated.
KEYWORDS: 14C analytics, carbon nanotubes, nanocomposites, N- and S-doping, reaction center protein.
INTRODUCTION
R e c e n t d e v e lo p m e n ts in in fo rm a tio n /c o m p u te r te ch n o lo g ies, in fo rm a tic s, o p to e le c tro n ic s a n d m o le c u la r b io lo g y m a d e it p o ssib le to m a n ip u la te m a te ria ls o n n a n o m e te r scales, o r even a t th e level o f m o lecu les, w h ich led to new disciplines o f th e 2 1 st ce n tu ry , su ch as “ N a n o te c h n o lo g y ” (see e.g. B h u s h a n 2004; W o lf 2004), “ N a n o - b io n ic s ” (see e.g. W o n g e t al. 2017; G ir a ld o e t al.
2014), “ (B io -)p h o to n ic s” (see e.g. W ilso n e t al. 2005; G e rd 2016; K n e ip p 2017), etc. T h a n k s to th e a c h ie v em e n ts o f th e se te ch n o lo g ie s new ty p e s o f m a te ria ls, n a n o m a te ria ls , are p ro d u c e d , w h ich offer new g e n e ra tio n s o f p ra c tic a l a p p lic a tio n s (S hoseyov a n d L ev y 2008;
C a rm e li e t al. 2007; D a r d e r e t al. 2007; F á b iá n e t al. 2010; S choles e t al. 2011; M a g y a r e t al.
2013; N a g y e t al. 2014).
A m o n g th e n a n o m a te ria ls , th e n a n o c o m p o s ite s are o f special in te re st b ec au se th e a d v a n ta g e o u s p ro p e rtie s o f th e c o m p o n e n ts c a n be c o m b in e d a n d /o r new c h a ra c te ristic s c a n a p p e a r w ith th e p o ssib ility o f tu n a b le , p red e fin e d m a n n e r (K a m ig a ito 1991; E v a n g e lo s 2007; N e m e th e t al.
2017). T h e special class o f n a n o c o m p o s ite s is th e b io -n a n o c o m p o site s, w h ich are co n sid ered as m a te ria ls o f th e fu tu re ( D a rd e r e t al. 2007; S ho sey o v a n d L ev y 2008; R u iz -H itz k y e t al. 2010;
N a g y e t al. 2014). A lth o u g h th e e x p lo ita tio n o f th e b io -n a n o c o m p o site m a te ria ls is m a in ly a w ish a t p re se n t, th e re is in te n siv e re se a rc h in m a n y la b o ra to rie s aim in g to ex p lo re th e ir fu n d a m e n ta l p ro p e rtie s o r to d e v e lo p th e ir p o ssib le p ra c tic a l a p p lic a tio n s. T h e ex tre m ely h ig h specificity, sensitivity, a n d effectiveness o f th e b io lo g ica l c o m p o n e n t o ffer p ro m isin g a p p lic a tio n s e.g. in activ e e lem e n ts o f b io se n so rs (Li e t al. 2011; C resp ilh o 2013; H o u e t al. 2016; S zabó e t al. 2017), in te g ra te d o p tic a l ( F á b iá n e t al. 2010, 2011), a n d o p to e le c tro n ic ( F la n a g a n e t al.
1988) devices. M a n y a tte m p ts h a v e b e e n m a d e fo r c re a tin g lig h t energy c o n v e rtin g (e.g. p h o to v o lta ic ) b io -n a n o c o m p o site devices (K ie tz k e 2007; Lee e t al. 2014; H a r tm a n n e t al. 2014;
S zekeres e t al. 2015; S z ab ó e t al. 2015). A d d itio n a lly , th e p ro ce sses h a p p e n in g a t th e in te rfa ce
*Corresponding author. Email: szabo.tibor@med.u-szeged.hu.
2 T Szabo et al.
b etw e en th e b io lo g ic a l a n d n o n -b io lo g ic a l c o m p o n e n ts c a n be a useful m o d e l to av o id
u n w a n te d re a c tio n s a g a in st th e h isto c o m p a tib ility o f artificial tissues, o rg a n s o r o th e r im p la n ts 44
in living beings. 45
T o im prove th e electrical properties o f M W C N T s, d o ping o f lattice o f c a rb o n n an o tu b es w ith 46
Q 5 different h etero ato m s (N , B, o r S) (D uclaux 2002; L iu et al. (2017); S h arm a e t al. (2017) via chem ical 47 Q 6 v ap o r deposition technique is a very p o p u la r m e th o d in th e field o f c a rb o n n an o tu b e science. 48 T h e re are several fields w h ere iso to p e a n a ly tic s a n d r a d io a n a ly tic s p ro v id e useful in sig h ts to 49 design, c re a te a n d ap p ly n a n o m a te ria ls (also (b io -)n a n o c o m p o site s) in e n v iro n m e n ta l sciences 50 as w ell ( G e r a n io e t al. 2010; F ei e t al. 2012; K i m e t al. 2 0 1 3 ,2 0 1 4 ; M a g y a r e t al. 2013; R o i g e t al. 51 2016; Y e d r a e t al. 2016; G o ttse lig e t al. 2017). E x a m p le s are as follow s: 52 I. D e te r m in a tio n o f ty p e s/so u rce s o f p re c u rso rs ( c a rb o n so u rces fro m d iffe re n t e n v iro n m e n t), 53 h e te ro a to m s (e.g. N o r S c o n te n t) w h e n c a r b o n m a te ria ls a re p ro d u c e d . 54 II. D e te r m in a tio n o f elem e n t c o n te n t vs. p h y sic o -/ch e m ic al c h a ra c te ristic s o f ( c a rb o n b a se d as 55 w ell) n a n o m a te ria ls . W e are fo cu sin g o n th e effects o f d o p in g (N , S d o p in g ), c o v e rin g w ith 56 m e ta l oxides o r fu n c tio n a liz a tio n o n th e o p tic a l a n d electric p ro p e rtie s. 57 III. F u n c tio n a liz a tio n w ith b io lo g ic a l m a te ria ls le a d s to th e c re a tio n o f b io -n a n o h y b rid 58 m a te ria ls w h ich are m a te ria ls o f th e fu tu re . Is o to p e a n a ly tic s h elp s in d e te rm in a tio n o f 59 yield o f fu n c tio n a liz a tio n , a b s o lu te q u a n tity o f c o m p o n e n ts a n d en z y m e activity. 60 O u r aim s w ere to p re p a re n itro g e n a n d su lfu r d o p e d c a rb o n n a n o tu b e s a n d to d e te rm in e th e 61 h e te r o a to m a n d 14C c o n te n t fo r fu rth e r c h a ra c te riz in g th e b io h y b rid m a te r ia l fo r fu tu re use. 62 T h e h e te r o a to m c o n ta in in g M W C N T s will be used as c a rrie rs a n d re a c tio n c e n te r p ro te in s 63
(R C s) as sensitizer m a tric e s. 64
MATERIALS AND METHODS 65
Preparation of Nitrogen and Sulfur Doped CNTs 66
D u rin g th e e x p e rim e n ts e ith e r F e (III)-C o (II)/c a lc iu m c a r b o n a te o r n ic k el(II)-o x id e c a ta ly sts 67 w ere used. In th is stu d y , a c a ta ly s t c o n ta in in g 5 m /m % F e a n d 5 m /m % C o w as p re p a re d b y 68 th e im p re g n a tio n te c h n iq u e . C a lc u la te d a m o u n ts o f C o (II)-a c e ty la c e to n a te , F e (III)- 69 a c e ty la c e to n a te a n d C a C O 3 w ere m ix e d in a b e a k e r w ith distilled w a te r. A fte r th a t a sh o rt 70 so n ic a tio n p ro ce ss w as ap p lie d to p re v e n t th e a g g re g a tio n o f solid p re c u rso r p article s. 71 A m m o n ia w as a d d e d to th e system to set th e p H to 9, a n d th e n th e d isp e rsio n w as p la ce d o n a 72 m a g n e tic stirre r a n d w as h e a te d a n d stirred in te n se ly a t 70°C u n til m o s t o f th e so lv e n t h a d 73 e v a p o ra te d . A fte r th e e v a p o ra tio n , th e p o w d e r c a ta ly st w as d rie d a t 100°C fo r 24 h r. In th e case 74 o f th e n ic k el(II)-o x id e c a ta ly st, c a lc u la te d a m o u n t o f N iO w as d issolved in a c e to n e a n d th e 75
su sp e n sio n w as d r o p p e d o n to a Si-sheet. 76
In this study, cataly tic chem ical v a p o r d ep o sitio n (C C V D ) m e th o d w as applied to synthesize 77 n itro g e n a n d sulfur d o p e d m u ltiw alled c a rb o n n a n o tu b e s (M W C N T s). N itro g e n a n d h y d ro g en 78 gases w ere passed th ro u g h th e re a c to r to m a in ta in a n in e rt a tm o sp h e re d u rin g the synthesis th a t 79 prev en ts M W C N T s fro m o x id a tio n a t h igher te m p eratu re s. T h e elem ental n itro g e n gas did n o t 80 ta k e p a r t in th e d o p in g process itself. T herefore, acetylene gas, th io p h e n e, a n d trip ro p y la m in e 81 (T P A ) w ere used as c a rb o n , sulfur a n d n itro g e n precursors, respectively. A cetylene a n d n itro g e n 82 gases w ere in tro d u c e d in to th e system b y passing th ro u g h a Y -sh ap ed ju n c tio n , w hile th e liquid- 83 p h ase T P A o r th io p h e n e w ere ad d e d b y th e b u b b lin g technique, w here the gases w ere co n d u c te d 84
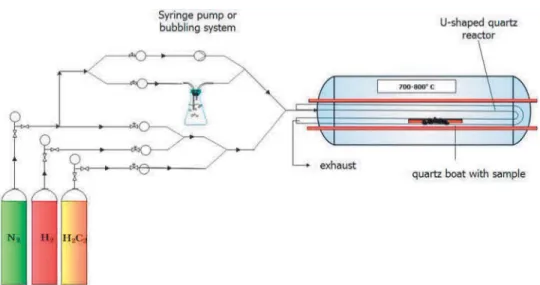
Syringe pump or bubbling system
- * o —
exhaust
quartz boat with sample
Figure 1 Schematic image of the CCVD reactor
in to liquid T P A o r th io p h e n e, b efore the rea cto r, th u s th e gas b ubbles carried th e T P A o r th io ph en e to th e rea ctio n site (F igure 1). F o r m o re intense T P A feed, in jection m e th o d using a syringe p u m p w as also applied; fo r fu rth e r exp erim en tal details see Szekeres e t al. (2015).
In these ex p e rim en ts, 150 m g o f c a ta ly st w as m e a su re d in to a q u a rtz b o a t. A fte r 15 m in o f leach in g , th e q u a r tz b o a t w as p la c e d in to th e o v en h e a te d u p to 720°C . In th e follo w in g step, a fte r th e r e a c to r w as h e a te d , ac ety len e flow w as set to 35 L h r-1 a n d th e M W C N T g ro w in g p ro ce ss sta rte d . T o finalize th e sy nthesis, th e ac ety len e flow ra te w as set to zero , a n d a fte r a sh o rt-tim e o f le ac h in g , th e re a c to r w as co o led d o w n to ro o m te m p e ra tu re to co llect th e M W C N T sam ples. I n all cases T P A o r th io p h e n e w as fed in to th e re a c to r d u rin g th e w hole re a c tio n tim e (30 m in).
Preparation of MWCNT/RC Complexes
RC Purification
T h e c a ro te n o id le ss R -2 6 s tra in o f R h o d o b a c te r ( R b . ) s p h a e r o id e s p u rp le b a c te riu m w as g ro w n p h o to -h e te ro tro p h ic a lly u n d e r a n a e ro b ic c o n d itio n s in a S istro m -m e d iu m su p p le m e n te d w ith p o ta ssiu m su c cin a te (S istro m 1960). R C s w ere p re p a re d by L D A O (la u ry ld im e th ila m in e N -o x id e , F lu k a ) so lu b iliz a tio n a n d s ta n d a r d p r o te in p u rific a tio n m e th o d s (a m m o n iu m su lfate p re c ip ita tio n , D E A E S ep h acell (S igm a) c o lu m n c h ro m a to g ra p h y a n d u ltra filtra tio n ) as d escrib ed p rev io u sly (M a r o ti a n d W r a ig h t 1988).
Functionalzation of MWCNTs with -CO O H Group
M W C N T s w ere fu n c tio n a liz e d w ith ca rb o x y l g ro u p s in a q u e o u s n itric acid so lu tio n w ith a c o n c e n tra tio n o f 1 0 m /m % fo r 1 hr. A fte r fu n c tio n a liz a tio n , th e sa m p le s w ere w a sh e d w ith distilled w a te r a n d p u t in a d rie r a t 90°C fo r 2 hr.
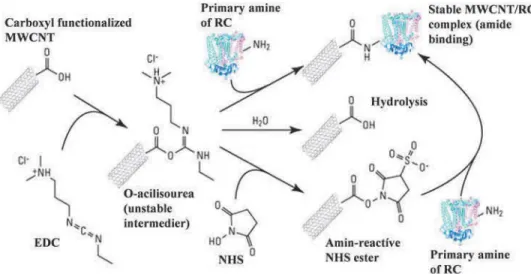
Binding RC to MWCNTs
R C w as b o u n d to c a rb o x y l-fu n c tio n a liz e d M W C N T s by E D C /N H S ch e m istry p ro c e d u re . C N T w as a c tiv a te d b y th e a d d itio n o f c ro sslin k ers N -h y d ro x y su c c in im id e (N H S ) a n d
85 86 87 88 89 90 91 92 93 94 95 96 97 98 99 100 101
102 103 104 105
106 107 108
4 T Szabó et al.
Primary amine of RC
Stable MWCNT/RC complex (amide
; binding) Carboxyl functionalized
MWCNT
0 Hydrolysis
O -a c il iso u r e a
(unstable intermedier)
Amin-reactive
NHS ester Primary amine
of RC
Figure 2 Schematic representation of binding reaction center to -CO O H functionalized MWCNTs by the EDC/
NHS method.
l-[3 -d im e th y la m in o p ro p y l]-3 -e th y l-c a rb o d iim id e (E D C ). A fte r a c tiv a tio n , th e m ix tu re w as d ia ly ze d in p o ta s s iu m p h o s p h a te b u ffe r (O .lM , p H 7.0) to re m o v e th e u n b o u n d cro sslin k ers.
T h e n , th e c a lc u la te d a m o u n t o f R C so lu tio n (ty p ically ca. l0 0 p M ) w as a d d e d to th e ac tiv a te d M W C N T a n d it w as stirre d a t 4 °C fo r 3 hr. F in a lly , th e sa m p le w as se p a ra te d a n d w ash e d by an u ltra c e n trifu g e u n til th e ste a d y -sta te a b s o rp tio n sp e c tru m o f th e s u p e rn a ta n t d id n o t show th e c h a ra c te ristic p e a k s o f th e R C in th e n e a r in fra re d . T h e re a c tio n schem e is su m m a riz e d in F ig u re 2.
Electron Microscopy
E a c h sa m p le w as c h a ra c te riz e d b y tra n sm issio n e le c tro n m ic ro sc o p y (T E M , F E I , T e c h n a i G 2 20 X -T W IN , 200kV ) to stu d y th e ir m o rp h o lo g y , w h ich p la y s a key ro le in th e u n d e rs ta n d in g o f th e M W C N T d o p in g . T h e sa m p le s w ere p re p a re d as follow s: first a sm all a m o u n t o f sa m p le w as d isp ersed in a b s o lu te e th a n o l b y u ltra s o n ic a tio n (35 k H z) in a n u ltra s o n ic a tio n b a th (T ra n sso n ic T 5 7 0 /H ), th e n a few d ro p s o f th e d isp e rsio n w ere p la c e d o n a 200 m e sh C u T E M g rid w ith c a r b o n layer.
X-Ray Powder Diffractometry (XRD)
T h e g ra p h itic p ro p e rtie s o f M W C N T s w as in v e stig a ted b y X R D m e th o d (R ig a k u M iniflex I I D iffra c to m e te r) utilizin g c h a ra c te ristic X -ra y (C u , Kot) r a d ia tio n (angle ra n g e 0 = 2 0 -7 0 °).
Raman Spectroscopy
G ra p h itic p ro p e rtie s o f M W C N T s w ere also verified b y R a m a n sp e ctro sc o p y m e a su re m e n ts (T h e rm o Scientific D X R R a m a n m ic ro sc o p e ) usin g 532 n m la se r e x c ita tio n , 2 0 x objective,
l 0 m W la ser p o w e r a n d l 5 m i n m e a su rin g tim e Measurements of the Radiocarbon Contents
T h e c a r b o n c o n te n t o f th e sa m p le s w as lib e ra te d b y sealed tu b e c o m b u s tio n m e th o d . T h e sam p le a n d th e M n O 2 o x id a n t w ere w eig h ted in to a g lass tu b e . W h e n th e in itia l sa m p le w as in a liq u id m a trix it w as w eig h ted in to th e c o m b u s tio n tu b e a n d d rie d w ith th e h e lp o f a freeze d ry er
109 110 111 112 113 114 115 116 117 118 119 120 121 122 123 124 125 126 127 128 129 130 131 132
u n it. T h e q u a n tity o f th e sa m p le w as c a lc u la te d fro m th e c o m b u stio n tu b e w ith th e d rie d sam p le
a n d th e e m p ty m a ss o f th a t. T h e tu b e s w ere e v a c u a te d to < 5 .1 0 _3 m b a r a n d sealed by a to rc h . 133 T h e sa m p le s w ere c o m b u ste d to C 0 2 in a m uffle fu rn a c e a t 550°C fo r 48 h r. T h e g a in e d C 0 2 w as 134 pu rified using a d e d ic a te d g as h a n d lin g system eq u ip p e d w ith cry o g en ic tr a p s in o rd e r to rem o v e 135 th e o th e r c o m b u ste d g a s c o m p o n e n ts . T h e q u a n tity o f th e p u re C 0 2 w as d e te rm in e d in a k n o w n 136 v o lu m e b y h ig h -p re c isio n p ressu re se n so r (Jan o v ic s 2016). T h e yield o f th e c a r b o n e x tra c tio n 137 c a n be c a lc u la te d fro m th e q u a n tity o f th e p u re C 0 2. T h e tr a p p e d a n d c lea n ed C 0 2 w ere 138 co n v e rte d to g ra p h ite by zinc re d u c tio n sealed tu b e g ra p h itiz a tio n m e th o d (R in y u e t al. 2013; 139 0 r s o v s z k i a n d R in y u 2015). In th e case o f less th a n 100 m ic ro g ra m s c a rb o n , zinc m ic ro - 140
g ra p h itiz a tio n te c h n iq u e w as used (R in y u e t al. 2015). 141
T h e m e a su re m e n ts o f th e 14C c o n te n ts w ere c a rrie d o u t o n a M I C A D A S ty p e a c c e le ra to r 142 m a ss s p e c tro m e te r (S ynal e t al. 2004, 2007) in th e In s titu te fo r N u c le a r R e se a rc h , D e b re c e n , 143 H u n g a ry (M o ln a r e t al. 2013). In o rd e r to tr a c k p o ssib le m o d e rn c a r b o n c o n ta m in a tio n 144 d u rin g th e p r e tr e a tm e n t a n d c o m b u stio n p ro cess, we h a v e e x tra c te d c h e m ic al s ta n d a rd s w ith 145 w ell-k n o w n 14C a c tiv ity (IA E A C 7 a n d C8; L e C le rc q e t al. 1998) o n th e sa m e tr e a tm e n t line 146 a n d m e a su re d th e m to g e th e r w ith th e sa m p le s in th e sam e m e a su re m e n t m a g az in es. B A T S 147 A M S d a t a e v a lu a tio n so ftw a re w as used to tre a t, p ro ce ss a n d an a ly z e all o f th e 14C d a t a 148
(L u k a s 2010). 149
Measurement of Stable Isotope Ratios 150
S tab le iso to p es w ere m e a su re d b y a T h e rm o F in n ig a n D e lta plusX P iso to p e r a tio m a ss spec- 151 tro m e te r a tta c h e d to a n e le m e n ta l a n a ly z e r (F is o n s N A 1 5 0 0 N C S ). T h is E A -I R M S m e th o d is 152 b ased o n th e ra p id o x id a tio n o f th e sa m p le b y flash c o m b u stio n , w h ich c o n v e rts all th e o rg a n ic 153 a n d in o rg a n ic su b sta n c e s in to c o m b u stio n p ro d u c ts, th e n th e re su lte d gases are se p a ra te d in a 154 c h ro m a to g ra p h ic c o lu m n a n d d e te c te d b y th e m a ss sp e c tro m e te r (M a jo r e t al. 2017). T h e 155 m e a su re d v alu es are expressed in d e lta n o ta tio n like 8 13C a n d 8 15N , w h ich d e lta v alu es are 156 de n ed as follow s: 8 ( I ) = (R / R - 1) x 1000, w h ere R is th e 13C /12C , 15N / 14N 157 ra tio in th e sam p le o r in th e in te rn a tio n a l referen ce m a te ria l as in d ic a te d . T h e u n c e rta in ty o f th e 158
m e a su re m e n ts is 0 . 2 I fo r S 13C a n d ± 0 . 3 I fo r 8 15N . 159
Measurement of Sulfur Content 160
C a rb o n n a n o tu b e sa m p le s w ere dig ested usin g a M a rs 5 m ic ro w a v e system . 50 m g o f th e sa m p le 161 w as w eig h ted in to th e T eflo n b o m b s a n d w as dig ested by 2 m L 67% (m /m ) n itric acid (su p ra - 162 p u re , V W R C hem icals). T h e ap p lie d p o w e r w as 800 W , 2 0 0 °C w as re a c h e d w ith in 20 m in , a n d 163 it w as h e ld fo r 30 m in . T h e sa m p le s w ere tra n s fe rre d in to 50 m L v o lu m e tric flasks a n d filled u p 164
w ith u ltra p u re w ate r. 165
T h e an a ly sis o f su lfu r w as p e rfo rm e d b y A g ile n t 8800 IC P -Q Q Q -M S system , u sin g M S /M S 166 m o d e . C h e m ic a l re a c tio n cell (C R C ) w as o p e ra tin g w ith o xygen re a c tio n gas. T h e su lfu r w as 167 m e a su re d w ith m a ss-sh ift, m e a su re d o n m /z 48 (cf. E q u a tio n 1): 168
32S + + 32 0 2 = 32S 160 + + 160 (1)
RESULTS AND DISCUSSIONS 169
Structural Characterizations of MWCNTs 170
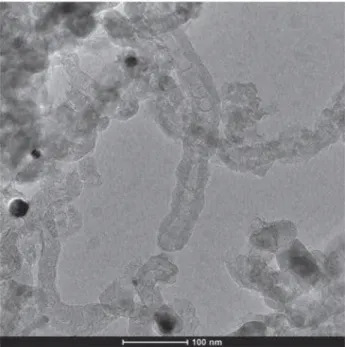
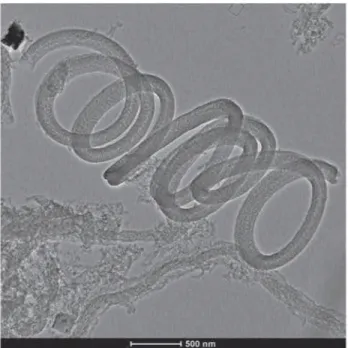
By c o m p a rin g th e T E M im ag es o f u n d o p e d (F ig u re 3), N -d o p e d (F ig u re 4) a n d S -d o p e d 171 (F ig u re 5), m o rp h o lo g ic a l c h a n g e s c a n be clea rly identified. T h e m ic ro g ra p h o f N -d o p e d 172 M W C N T s (F ig u re 4) rev e ale d th a t th e re a c tio n p r o d u c t w as a c a r b o n d e p o s it c o n ta in in g
6 T Szabo et al.
Figure 3 Representative TEM image of undoped CNTs.
Figure 4 Representative TEM image of N-doped MWCNTs, visualizing the structural degenerations caused by the dopant, e.g. the bamboo structure.
h o llo w M W C N T s w ith b a m b o o -lik e segm ents. D iffe re n t fro m th e N -d o p e d n a n o s tru c tu re s , S -d o p e d n a n o s tru c tu re s sho w ed a special coiled m o rp h o lo g y , w h ich is o v era ll re p re se n ta tiv e fo r th e sam ples. T h e stru c tu ra l v a rie tie s fo u n d in th e N -d o p e d a n d S -d o p e d sa m p le s ca n
173
Figure 5 Representative TEM image of S-doped MWCNTs, showing the coiled morphology.
Figure 6 XRD diffractograms of undoped and N-doped CNTs.
p o ssib ly in d ic a te th e p rese n ce o f d o p a n ts , as sim ila r re a c tio n c o n d itio n s w ith d iffe ren t
h e te r o a to m resu lted in d istin c t s tru c tu ra l ch an g es. T h e stru c tu ra l d ifferen ces c a n re su lt fro m th e 174 m a n y fo rm s th e d o p a n ts c a n in c o rp o ra te in to th e M W C N T la ttic e (Szekeres e t al. 2015; Y a n g 175
e t al. 2015). 176
P o w d e r X R D m e a su re m e n ts w ere p e rfo rm e d to d e te rm in e , w h e th e r th e sy n th esized sa m p le s 177 possess a g ra p h itic stru c tu re . T h e d iffra c to g ra m s in F ig u re 6 show th e c h a ra c te ristic reflec tio n s 178 o f th e g ra p h itic la ttic e a t 20 = 26.11° a n d 44.65°, b u t w ith d iffe re n t rela tiv e in te n sities to e a ch 179 o th e r in th e tw o d a ta se ts, w h ich is th e re su lt o f th e significantly c h a n g e d stru c tu re s. T h e resu lts, 180 h o w ev e r, assu re th a t th e severe s tru c tu ra l d e g e n e ra tio n u p o n d o p in g d o es n o t n ecessarily re su lt 181
8 T Szabo et al.
uridaped MWCNT --- N-doped MWCNT
0.66 -
0.44 -
0.00 -
500 1000 1500 2000 2500 3000 3500 Raman shift (cm’ )
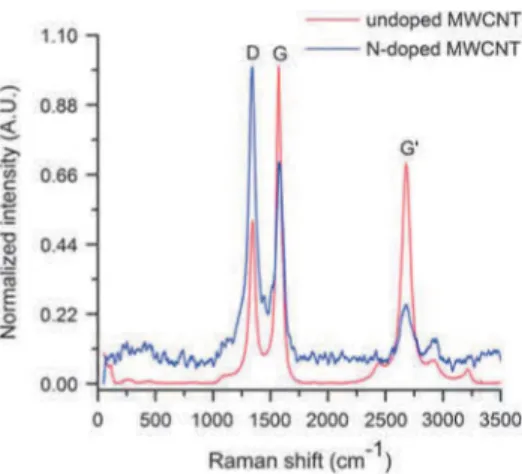
Figure 7 Raman spectra of undoped and N-doped CNTs.
in th e loss o f g ra p h itic p ro p e rtie s, a n d th e re fo re in th e exclu sio n o f u n iq u e electrical ch a racteristics.
R a m a n sp e c tra o f u n d o p e d a n d d o p e d M W C N T s a re sh o w n in F ig u re 7. G ra p h itic p ro p e rtie s are o fte n e x p lain e d b y th e in te n sity ra tio s o f th e D -b a n d (1341 cm -1 , assig n ed to th e p resen ce o f d efe ct sites) a n d th e G -b a n d (1527 c m 1, assig n ed to th e g ra p h itiz a tio n level). T h e in c re ase d D -b a n d in te n sity , as w ell as th e d e c re m e n t in th e G -b a n d in te n sity su g g est th a t th e c o n tin u o u s g ra p h itic la ttic e is significantly c o rru p te d in th e N -d o p e d sam ple. H o w e v e r, th e p resen ce o f th e G - a n d G ’-b a n d s in d ic a te th a t th e sam p le d id n o t lose its o v era ll g ra p h itic id e n tity , o n ly its h o m o g e n e ity — d u e to th e a lte re d re a c tio n c o n d itio n s c a u se d b y in tro d u c in g a d o p a n t— is low er.
S im ilar c o n c lu sio n s c a n be d ra w n fro m th e X R D a n d R a m a n in v e stig a tio n s o f th e S -d o p ed M W C N T s.
Isotope Analytical Investigations of CNTs
N in e d iffe re n t ty p e o f c a r b o n n a n o tu b e s w ere an a ly z e d b y iso to p e a n a ly tic a l m e th o d s a n d th e sam p le id e n tific a tio n w ith c h a ra c te ristic s (T a b le 1) a n d resu lts o f sta b le N a n d C c o n te n t (T a b le s 2, 3) a n d 14C c o n te n t (T a b le 4) are su m m a riz e d .
T h e m ass sp e ctru m o f the sam ple n a n o tu b e is co m p a re d to th e reference m a te ria l o f k n o w n c a rb o n a n d n itro g e n c o n te n t a n d all o f these calcu latio n s o f w eight p erc en t d e te rm in a tio n refer to the so-called signfiicant p eak . T h ere are sam ples w ith som e n itro g e n c o n te n t betw een 2.61% an d 5.3 m /m % (1/1466/9,10,13,14). T h e c a rb o n c o n te n t o f different sam ples is fairly different how ever, it ran g es betw een 33 a n d 67%. T h e sam ples w ith n itro g e n c o n te n t h a s a 8 15N value betw een 4 . 2 5 1 a n d 6 . 0 9 1 a n d th e stable c a rb o n iso to p e results cover a w ider ran g e betw een - 2 0 . 5 7 ! an d - 2 9 . 5 7 ! .
By using th io p h e n e , we fo u n d th a t th e in c re ase o f C V D synthesis te m p e ra tu re resu lted in h ig h e r c a r b o n d e p o s it th u s h ig h e r c a r b o n (m /m % ) (T a b le 2). O n th e o th e r h a n d , in c re asin g te m p e r a tu re is n o t a d v a n ta g e o u s fo r th e in c o rp o r a tio n o f h e te ro a to m s in to g ra p h itic stru c tu re s th e re th ro u g h th e b u ilt-in a m o u n t o f su lfu r is so m e w h a t lo w er a t 800°C .
T h e b u b b lin g m e th o d used fo r th e p r e p a ra tio n o f S -d o p e d C N T w as fo u n d to be m u c h less effective in th e sy n th esis o f N -d o p e d m a te ria ls, w hile u sin g T P A as d o p a n t. T h e sta b le N c o n te n t o f sam p les 11, 12, a n d 15 w as n o t d e te c ta b le (see T a b le 2), h o w ev e r, T E M
182 183 184 185 186 187 188 189 190 191 192 193 194 195 196 197 198 199 200 201 202 203 204 205 206 207 208
T a b le 1 Id e n tific a tio n a n d d e s c rip tio n o f d iffe re n t ty p e s o f n a n o tu b e s u sed fo r th e analysis.
S a m p le d e sc rip tio n
R e a c tio n tim e /
S am p le ID M e th o d L iq u id d o p in g tim e [min] T [°C]
I/1466/1 S -dop. B u b b lin g T h io p h e n e 30/30 700
I/1466/2 B u b b lin g T h io p h e n e 30 30 800
I/1466/9 N -d o p e d In je c tio n T P A - fe rro c e n e -a c e to n e 10 2 720
I/1466/10 In je c tio n T P A - fe rro c e n e -a c e to n e 20 5 720
I/1466/11 B u b b lin g T P A 40 40 720
I/1466/12 B u b b lin g T P A - a c eto n e 40 40 720
I/1466/13 In je c tio n T P A 20 5 720
I/1466/14 In je c tio n T P A 20 10 720
I/1466/15 B u b b lin g T P A 40 40 750
T a b le 2 S tab le n itro g e n , su lfu r a n d c a r b o n iso to p e resu lts o f in v e stig a te d n a n o tu b e species.
T h e sa m p le I D refers to th e sa m p le s listed in T a b le 1.
Sample ID
N ( 1 , A IR)
o ( 1 , A IR)
N itrogen (m/m%)
C ( 1 , VPDB)
o
( 1 , VPDB)
C arbon
(m m%) S ((mg/kg) o ((mg/kg)
I/1466/1 — — — -2 6 .7 9 0.16 33.30 318 3
I/1466/2 — — — -2 5 .8 9 0.04 41.94 256 4
I/1466/9 6.09 0.03 5.30 -2 4 .2 2 0.09 66.43 — —
I/1466/10 4.25 0.33 2.61 -2 7 .5 5 0.11 66.35 — —
I 1466 11 — — — -2 5 .0 5 0.16 45.41 — —
I/1466/12 — — — -2 6 .0 3 0.24 41.34 — —
I/1466/13 5.13 0.01 2.84 -2 9 .5 7 0.08 58.79 — —
I/1466/14 5.96 0.39 3.47 -2 9 .5 0 0.27 55.78 — —
I/1466/15 — — — -2 0 .5 7 0.04 37.56 — —
T a b le 3 14C c o n te n t o f th e in v e stig a te d n a n o tu b e s.
T h e sa m p le I D refers to th e sam p les listed in T a b le 1.
S am p le ID
C o m b u s tio n yield (%)
14C c o n te n t (p M C )
I/1466/9 56.0 18.0
I/1466/10 44.0 6.2
I/1466/11 63.0 4.0
I/1466/12 73.0 3.0
I/1466/13 59.0 13.6
I/1466/14 58.0 13.4
I/1466/15 33.0 34.1
Q7
in v e stig a tio n s rev e ale d som e b a m b o o -lik e c h a r a c te r o f th e se c a r b o n n a n o tu b e s , to o . S am p le 15
d e m o n s tra te d th a t in c re a sin g re a c tio n te m p e ra tu re is n o t a so lu tio n fo r g e ttin g h ig h e r N -d o p - 209 ing. F o r th e sy n th esis o f sam p le 12 th e liq u id used fo r b u b b lin g w as c h a n g e d , n a m e ly T P A w as 210 m ix e d w ith a c e to n e (1:9 r a tio ), w h ich p ro v id e d b e tte r v o la tility . H o w ev er, th is m o d ific a tio n d id 211
n o t re su lt in h ig h e r N -d o p in g eith er. 212
10 T Szabo et al.
T a b le 4 S u m m a ry o f d a ta o b ta in e d d u rin g d e te rm in a tio n o f th e 14C q u a n tity . S istro m m e d iu m is th e g ro w in g m e d iu m o f th e b a c te ria , R C is re a c tio n c e n te r p ro te in , f -M W C N T COOH is th e - C O O H fu n c tio n a liz e d 1/1466/11 sa m p le , a n d f -M W C N T COOH/R C is th e c a r b o n n a n o tu b e / re a c tio n c e n te r n a n o c o m p o site , respectively.
S am ple
S am p le q u a n tity (m g)
M e a su re d c a rb o n (m g)
Y ield
(%) p M C
S istro m -m e d iu m 9.93 2.07 21% 31.9 ± 0.5
R C 2.78 1.49 54% 66.1 ± 0.3
f-M W C N T 1.15 0.85 74% 6.4 ± 0 .1
f -M W C N T COOH/R C F
0.39 38.0 ± 2.0%
0.28 73% 29.1 ± 0 .1
A re a so n a b le e x p la n a tio n fo r th is sig n ifican t d iffe ren ce b etw e en th io p h e n e a n d T P A d o p a n ts c a n be fo u n d in th e ir v a p o r p ressu re v alu es a t ro o m te m p e ra tu re , w h ich d iffer b y 2 o rd e rs o f m a g n itu d e (T P A : 1.51 m m H g , th io p h e n e : 79.7 m m H g ). T h is ex p erien ce in sp ire d us to so m e w h a t m o d ify o u r C V D se tu p a n d b u ild a syringe p u m p in to th e system .
N -d o p e d sam ples p re p a re d w ith th e injection tech n iq u e (9, 10, 13, 14) resulted in signfficantly h igher stable N c o n te n t o f C N T sam ples (T able 2). I f we co m p a re th e N m /m % d a ta fo r sam ples 13 a n d 14, we c a n conclu d e th a t increasing d o p in g tim e fro m 5 to 10 m in (applying 20 m in rea ctio n tim e) c a n resu lt in a slightly h igher a m o u n t o f in c o rp o ra te d N . I t is k n o w n fro m literary d a ta (Y a d av e t al. 2005) th a t ferrocene is able to b lo c k the n itro g e n in c o rp o ra tio n in to th e c a rb o n n a n o tu b e structure, th e re fo re sam ples 9 a n d 10 w ere p re p a re d w ith ferrocene feed. S table iso to p e m easu rem en ts also revealed th a t th e in h ib itio n effect o f ferrocene a p p e ars only after a n in d u c tio n p eriod. 2 m in “T P A - ferrocene - a c eto n e” feed is n o t sufficient to b lo c k n itro g e n in c o rp o ratio n , hence th e stable N co n te n t value fo r sam ple 9 is sim ilar to th a t o f o th e r sam ples p re p a re d by injection. In creasin g ferrocene injection to 5 m in , th e block ag e c a n be alread y d etected (approx.
50% low er N -c o n te n t in T ab le 2) w hich is in ac co rd an c e w ith fo rm e r E M observations.
T h e 14C c o n te n t o f th e n a n o tu b e s w as also e x a m in e d a n d th e resu lts a re su m m a riz e d in T a b le 3. W e d id n o t g e t a v alid re s u lt in tw o sa m p le s (I/1466/1 a n d 2). T h e c o m b u stio n efficiency flu c tu a te s significantly fo r th e d iffe re n t sam ples. I t seem s th a t th is efficiency is h ig h ly d e p e n d e n t o n th e c a r b o n c o n te n t o f th e sam p le, w h ich is d e te rm in e d b y th e ap p lie d c a rrie rs a n d p re c u rso rs.
R e su lts in d ic a te th a t th e 14C c o n te n t o f th e n a n o tu b e s p ro d u c e d b y th e v a rio u s p r e p a ra tio n m o d e s v arie s w idely (fro m 3.0 p M C u p to 34.1 p M C ). Iso to p ic a n a ly tic a l studies h av e sh o w n th a t th e used sta rtin g m a te ria ls, p re c u rso rs a n d c a rrie rs h av e a stro n g iM u e n c e on th e c h a ra c te ristic s o f th e p ro d u c e d n a n o tu b e s . F o r b in d in g stu d ies, it is im p o r ta n t fo r th e 14C c o n te n t o f th e n a n o tu b e a n d th e p ro te in h a s to be significantly d iffe ren t. A s th e 14C c o n te n t o f th e in v e stig a te d p ro te in is d e te rm in e d by th e c a r b o n so u rce fro m w h ich th e p r o te in is d ev elo p ed , one asp ec t w as th e selectio n o f th o se n a n o tu b e ty p e s, w h ich h a v e th e lo w est 14C c o n te n t d u rin g th e analysis. E v e n tu a lly , th e tw o sa m p le c a n d id a te s fo r fu rth e r in v e stig a tio n s w ere I/1466/11 a n d I/1466/12 (cf. T a b le 4). W e used sa m p le I/1466/11 fo r n a n o c o m p o s ite p re p a ra tio n . A low 14C c o n te n t o f th e n a n o -tu b e is re q u ire d in o rd e r to b e tte r d e te rm in a tio n o f 14C c o n c e n tra tio n ch a n g e c a u se d by re a c tio n c e n te r co m p lex b in d in g (w hich is rec en t). T h is c a n be used to m o re a c c u ra te ly d e te rm in e th e b o n d e d R C ra tio .
In th e case o f th e re a c tio n ce n te r, th e p rim a ry c a r b o n so u rce is su p p lied b y th e S istro m -m e d iu m (31.9 ± 0.5 p M C , cf. T a b le 4). H o w e v e r, th e re c e n t a tm o sp h e ric c a rb o n -d io x id e (a ro u n d
100 p M C ) as se c o n d a ry so u rce also iM u e n c e s th e 14C c o n te n t o f th e b a c te riu m cells.
213 214 215 216 217 218 219 220 221 222 223 224 225 226 227 228 229 230 231 232 233 234 235 236 237 238 239 240 241 242 243 244 245
Measurements on MWCNT/RC Hybrid System
F o r p re p a rin g M W C N T /R C b io h y b rid system M W C N T s fu n c tio n a liz e d b y - C O O H g ro u p s w ere used, h e re a fte r referre d as f -M W C N T COOH. I n o rd e r to d e te rm in e th e a b s o lu te q u a n tity o f th e R C in th e f -M W C N T COOH/R C c o m p lex th e a m o u n t o f th e p r o te in b o u n d to th e f -M W C N T w as d e te rm in e d . F o r th is re a so n , th e p M C u n it w as d e te rm in e d f o r th e R C , f -M W C N T a n d f -M W C N T /R C sam ples. T h e R C fra c tio n o f th e f -M W C N T / R C co m p lex is o b ta in e d b y 14C b a la n c e e q u a tio n . 14C c o n te n t o f th e tw o -c o m p o n e n t m ix tu re is defined as E q u a tio n 2:
p M Cf _ m w c n tcooh / r c = Fr c ■ ° M Cr c + ( 1 - Fr c) - p M Cf _ m w c n tcooh (2)
w here p M C RC, p M C a n d p M C is th e m e a su re d 14C c o n te n t o f th e R C , f -M W C N T a n d f -M W C N T /R C co m p lex , respectively. F RC is th e R C fra c tio n o f th e 14C c o n te n t o f th e f -M W C N T /R C co m p lex . T h e R C f ra c tio n o f th e m ix tu re is o b ta in e d a fte r th e re a rra n g e m e n t o f 14C b a la n c e e q u a tio n (E q u a tio n 3):
Fr c =p M C f - MWCNTcooh / RC _ p M C f _ m w c n t cooh p M C RC _ p M C f _ MWCNTc
100
% . (3)By u sin g th e d a t a o b ta in e d d u rin g th e d e te rm in a tio n o f 14C q u a n tity a n d su m m a riz e d in T a b le 4, Fr c = 38.0 ± 2.0% c a n be c a lc u la te d . F in a lly , th e to ta l R C /f -M W C N T r a tio a n d th e a m o u n t o f R C (m /m ) b y th e k n o w n m o le c u la r w eig h t (M W : 80 k D a , 48% c a r b o n c o n te n t) c a n be c a lc u la te d using th e R C fra c tio n c o n s ta n t a n d th e c a r b o n e x tra c tio n yields o f th e a p p r o p r ia te c o m p o n e n ts . C a r b o n c o n te n t w as e s tim a te d b y th e R C stru c tu re o f 2 W X 5 .p d b d e p o site d in th e B ro o k h a v e n P ro te in D a ta B a n k (h ttp s ://w w w .rc sb .o rg /p d b /h o m e /h o m e .d o ) a fte r o m ittin g th e w a te r m o lecu les. T h e c a lc u la te d a m o u n t o f th e p h o to sy n th e tic re a c tio n c e n te r b o u n d to M W C N T s fo u n d 53 m /m % R C w h en th e p r o te in b o u n d b y th e E D C - N H S ch em ical b in d in g m e th o d .
CONCLUSIONS
F o r s u m m a ry , we c a n c o n c lu d e th a t b y a p p ly in g a specific c h a n g e in th e e le m e n t c o m p o sitio n o f th e p re c u rs o rs d u rin g N W C N T sy n th e sis specific d o p in g c a n b e ac h ie v e d , fo r e x a m p le N a n d S c a n be in se rte d in th e C N T s tru c tu re . T h e yield o f th e d o p in g w as p ro v e d b y th e is o to p e a n a ly tic a l in v e stig a tio n s. D o p in g th e M W C N T s b y N a n d S re su lte d in specific s tr u c tu ra l c h a n g e s w ith th e a p p e a r a n c e o f “ b a m b o o -lik e ” o r sp ira l s tru c tu re s, resp ectiv ely .
T h e iso to p e c o n s titu tio n o f b io h y b rid m a te r ia ls p r e p a re d f ro m th e b a r e a n d /o r d o p e d c a r rie r C N T s a n d e n z y m e s p ro v e d th a t iso to p e a n a ly tic s is a u se fu l to o l f o r d e te rm in in g th e q u a n ti
ta tiv e b in d in g o f th e b io lo g ic a l m a te r ia ls to th e c a r rie r m a tric e s. A fte r d e te rm in in g th e q u a n tita tiv e a m o u n t o f th e en z y m e th e specific a c tiv ity o f th e sa m p le c a n b e d e te rm in e d . T h e a m o u n t o f th e p h o to s y n th e tic r e a c tio n c e n te r b o u n d to M W C N T s c a n b e d e te rm in e d in a sen sitiv e w a y a n d fo u n d 53 m /m % R C w h e n th e p r o te in b o u n d by th e E D C - N H S ch e m ic al b in d in g m e th o d . D e ta ile d in v e s tig a tio n o f th e effec t o f d iffe re n t c a r b o n so u rce s u n d e r th e g r o w th c o n d itio n s a re b e y o n d th e sc o p e o f th is p u b lic a tio n , h o w e v e r, th is c a n b e a m a tte r o f f u rth e r in v e stig a tio n .
246 247 248 249 250 251 252
253 254 255 256 257
258 259 260 261 262 263 264 265 266 267 268 269 270 271 272 273 274 275 276 277 278 279 280 281 282 283
12 T Szabó et al.
ACKNOWLEDGMENTS
T h e re se a rc h w as su p p o rte d b y th e E u ro p e a n U n io n a n d th e S ta te o f H u n g a ry , co -fin an ced b y 284 th e E u ro p e a n R e g io n a l D e v e lo p m e n t F u n d in th e p ro je c t o f G IN O P -2 .3 .2 .-1 5 -2 0 1 6 - 285 00009 “ I C E R ” a n d fro m th e N a tio n a l R e se a rc h , D e v e lo p m e n t a n d I n n o v a tio n ( N K F I ) F u n d 286 (O T K A P D 1 1 6 7 3 9 ), a n d K .N . a c k n o w led g e s th e fin a n c ia l s u p p o r t o f O T K A N N 1 1 4 4 6 3 287
REFERENCES
288Bhushan B. 2004. Handbook o f Nanotechnology.
Berlin: Springer.
Carmeli I, Frolov L, Carmeli C, Richter S. 2007.
Photovoltaic activity of photosystem I-based self- assembled monolayer. Journal o f American Chem.
Soc. 129:12352-3.
Collini E. 2017. Biophotonics: That quantum feeling.
Q 9 Nature Physics.
Crespilho FN. 2013. Nanobioelectrochemistry: from Implantable Biosensors to Green Power Genera
tion. Berlin: Springer.
Daliento S, Chouder A, Guerriero P, Massi Pavan A,
Q 1 0 Mellit A, Moeini R, Tricoli P. 2017. Monitoring, diagnosis, and power forecasting for photovoltaic fields: a review. International Journal o f Photo
energy 2017:1356851.
Darder M, Pilar Aranda P, Ruiz-Hitzky E. 2007.
Bionanocomposites: a new concept of ecological, bioinspired, and functional hybrid materials. Adv.
Mater. 19:1309-19.
Duclaux L. 2002. Review of the doping of carbon nanotubes (multiwalled and single-walled).
Carbon 40(10):1751-64.
Evangelos M. 2007. Nanocomposites: stiffer by design. Nature Materials 6(1):9-11.
Fábián L, Wolff EK, Oroszi L, Ormos P, Dér A. 2010.
Fast integrated optical switching by the protein bacteriorhodopsin. Appl. Phys. Lett. 97:023305.
Fábián L, Heiner Z, Mero M, Kiss M, Wolff EK, Ormos P, Osvay K, Dér A. 2011. Protein-based ultrafast photonic switching. Optics Express.
19:18861-70.
Fei Z, Rodin AS, Andreev GO, Bao W, McLeod AS, Wagner M, Zhang LM, Zhao Z, Thiemens M, Dominguez G, Fogler MM, Castro Neto AH, Lau CN, Keilmann F, Basov DN. 2012. Gate
tuning of graphene plasmons revealed by infrared nano-imaging. Nature 487:82-5.
Flanagan MT, Sloper AN, Ashworth RH. 1988. From electronic to opto-electronic biosensors: an engineer
ing view. Analytica Chimica Acta. 213:23-33.
Geranio L, Hommes G, Shahgaldian P, Wirth-Heller A, Pieles U, Corvini PFX. 2010.
Radio (14C)- and fluorescent-doubly labeled silica nanoparticles for biological and environ
mental toxicity assessment. Environmental Chemistry Letters 8:247-51.
Gerd K. 2016. Biophotonics - Concepts to Application.
Singapore: Springer.
Giraldo JP, Landry P, Faltermeier SM, McNicholas TP, Iverson NM, Boghossian AA, Reuel NF,
Hilmer AJ, Sen F, Brew JA, Strano MS. 2014. 24®
Plant nanobionics approach to augment photo- 241
synthesis and biochemical sensing. Nature Materials 242
13:400-8. 244
Gottselig N, Amelung W, Kirchner JW, Bol R, 344
Eugster W, Granger SJ, Hernändez-Crespo C, Q f346
Herrmann F, Keizer JJ, Korkiakoski M, Laudon 3413
H, Lehner I, Löfgren S, Lohila A, Macleod CJA, Q BB8
Mölder M, Müller C, Nasta P, Nischwitz V, Paul- 348
Limoges E, Pierret MC. 394
Hartmann V, Kothe T, Poller S, El-Mohsnawy E, 360
Nowaczyk MM, Plumere N, Schuhmann W, 301
Rogner M. 2014. Phys. Chem. Chem. Phys. 363
16:11436. 304
Hou S, Zhang A, Su M. 2016. Nanomaterials for 364
Biosensing Applications. Nanomaterials 6:58. 306
Janovics R. 2016. Development of radiocarbon-based 3013
measuring methods and their application for 368
nuclear environmental monitoring [PhD thesis, in 358
Hungarian]. University of Debrecen and Hun- 304
garian Academy of Sciences Institute for Nuclear 360
Research. https://dea.lib.unideb.hu/dea/handle/ 311
2437/217939?locale-attribute=en. 312
Jones MR. 2004. The petite purple photosynthetic Q t3Jb3
powerpack. Biochem Soc. Trans., 37:400-7. 314
Kamigaito O. 1441. What can be improved by nan- 316
ometer composites? J. Jpn. Soc. Powder Powder 366
Metall. 38:315-21. 317
Kietzke T. 2007. Recent Advances in Organic Solar Cells. 364
Adv. Opto Electron. Article ID 40285, 15 p. 374
Kim H, Osofsky M, Prokes SM, Glembocki OJ, 321
Pique A. 2013. Optimization of Al-doped ZnO 321
films for low-loss plasmonic materials at tele- 322
communications wavelengths. Appl. Phys. Lett. 323
102:171103. 324
Kim J, Naik GV, Shalaev VM, Gavrilenko AV, 326
Dondapati K, Gavrilenko VI, Prokes SM, Glem- 327
bocki OJ, Boltasseva A. 2014. Optical properties 378
of gallium-doped zinc oxide-a low-loss plasmonic 328
material: first principles theory and experiment. 324
Phys Rev X. 3(4):041037. 381
Kneipp J. 2017. Interrogating cells, tissues, and living 382
animals with new generations of surface-enhanced 383
Raman scattering probes and labels. A C S Nano. 384
11(2):1136-41. 385
Le Clercq M, van der Plicht J, Gröning M. 1448. New 386
14C reference materials with activities of 15 and 50 387
pMC. Radiocarbon 40(1):295-2. 338
Lee CW, Kim OY, Lee JY. 2014. Organic materials 338
for organic electronic devices. J. Ind. Eng. Chem. 334
20:1148-208. 340
Li S, Singh J, Li H, Ipsita A., Banerjee IA. 2011.
Biosensor Nanomaterials. Wiley-VCH Verlag GmbH & Co. KgaA.
Liu S, Li GZ, Gao YY, Xiao ZR, Zhang JF, Wang QF, Zhang XW, Wang L. 2017. Doping carbon nanotubes with N, S, and B for electrocatalytic oxygen reduction: a systematic investigation on single, double, and triple doped modes. Catalysis Science & Technology 7(18):4007-16.
Magyar M, Hajdu K, Szabó T, Endrődi B, Hernádi K, Horváth E, Magrez A, Forró L, Visy C, Nagy L.
2013. Sensing hydrogen peroxide by carbon nano- tube/horse radish peroxidase bio-nanocomposite.
Phys. Status Solidi B 250:2559-63.
Major I, Gyökös B, Túri M, Futó I, Filep Á, Hoffer A, Furu E, Jull AJT, M olnár M. 2017.
Evaluation of an autom ated EA-IRMS method for total carbon analysis of atmospheric aerosol at HEKAL. Journal o f Atmospheric Chemistry 75 (1): 8 5-96.
Maróti P, Wraight CA. 1988. Flash-induced H+
binding by bacterial photosynthetic reaction centers: Comparison of spectrophotometric and conductimetric methods. Biochim Biophys Acta.
934:314-28.
Molnár M, Rinyu L, Veres M, Seiler M, Wacker L, Synal HA. 2013. EnvironMICADAS: A mini 14C AMS with enhanced gas ion source interface in the Hertelendi Laboratory on Environmental Studies (HEKAL). Radiocarbon 55(2):338-44.
Nagy L, Magyar M, Szabo T, Hajdu K, Giotta L, Dorogi M, Milano F. 2014. Photosynthetic machineries in nano-systems. Current Protein &
Peptide Science 15:363-73.
Nemeth K, Kovacs L, Reti B, Belina K, Hernadi K.
2017. The synthesis and investigation of Si02- MgO coated multiwalled carbon nanotube/poly- mer composites. Journal o f Nanoscience and Nanotechnology 17(8):5445-52.
Orsovszki G, Rinyu L. 2015. Flame-sealed tube gra- phitization using zinc as the sole reduction agent:
Precision improvement of EnvironMICADAS 14C measurements on graphite targets. Radio
carbon 57(3):979-90.
Pilegaard K, Romano N, Sebastiá MT, Stahli M, Voltz M, Vereecken H, Siemens J, Klumpp E.
Q 1 3 2017. Elemental composition of natural nano
particles and fine colloids in European forest stream waters and their role as phosphorus car
riers. Global Biogeochemical Cycles 31(9).
Rinyu L, Molnár M, Major I, Nagy T, Veres M, Kimák Á, Wacker L, Synal HA. 2013. Optimiza
tion of sealed tube graphitization method for environmental 14C studies using MICADAS.
Nuclear Instruments and. Methods in Physics Research B 294:270-5.
Rinyu L, Orsovszki G, Futó I, Veres M, Molnár M.
2015. Application of zinc sealed tube graphitiza
tion on sub-milligram samples using Envi- ronMICADAS. Nuclear Instruments and Methods in Physics Research B 361:406-13.
Roig JL, Gómez-Vallejo V, Gibson PN. 2016. Iso- 452 topes in Nanoparticles: Fundamentals and. Appli- 492 cations. Singapore: Pan Stanford Publishing. 494 Ruiz-Hitzky E, Darder M, Aranda P. 2010. Progress 394 in bionanocomposite materials. In: Cao G, Zhang 456 Q, Brinker CJ, editors. Annual Review o f Nanor- 458 esearch. Singapore: World Scientific Publishing. p 458
149-89. 359
Scholes GD, Fleming GR, Olaya-Castro A, van 380 Grondelle R. 2011. Lessons from nature about 48®
solar light harvesting. Nature Chem. 3:763-74. 401 Sharma A, Dasgupta K, Patwardhan A, Joshi J. 2017. 402 Kinetic study of nitrogen doped carbon nanotubes 404 in a fixed bed. Chemical Engineering Science 405
170:756-66. 466
Shoseyov O, Levy I. 2008. Nanobiotechnology: Bio- 468 inspired. Devices and. Materials o f the Future. 468
Totowa: Humana Press. 469
Siström WR. 1960. A requirement for sodium in the 470 growth of Rhodopseudomonas spheroid.es. J Gen 471
Microbiol. 22:778-85. 471
Synal HA, Döbeli M, Jacob S, Stocker M, Suter M. 413 2004. Radiocarbon AMS towards its lower- 474 energy limits. Nuclear Instruments and Methods 414 in Physics Research B 223-224:339-45. 4T6 Synal HA, Stocker M, Suter M. 2007. MICADAS: A 477 new compact radiocarbon AMS system. Nuclear 478 Instruments and. Methods in Physics Research B 418
259:7-13. 419
Szabó T, Nyerki E, Tóth T, Csekő R, Magyar M, 421 Horváth E, Hernádi K, Endrődi B, Visy Cs, 481 Forró L, Nagy L. 2015. Generating photocurrent 422 by nanocomposites based on photosynthetic 423 reaction centre protein. Phys. Status Solidi B252 484
(11):2614-19. 486
Szabó T, Csekő R, Hajdu K, Nagy K, Sipos O, 487 Galajda P, Garab Gy, Nagy L. 2017. Sensing 427 photosynthetic herbicides in an electrochemical 488 ow cell. Photosynth. Res. 132(2):127-34. 420 Szabó T, Nyerki E, Tóth T, Csekő R, Magyar M, 430 Horváth E, Hernádi K, Endrődi B, Visy C, 431 Forró L, Nagy L. 2015. Generating photo- 432 current by nanocomposites based on photo- 433 synthetic reaction centre protein. Phys. Status 434
Solidi B 252:2614-19. 436
Szabó T, Magyar M, Hajdu K, Dorogi M, Nyerki E, 496 Tóth T, Lingvay M, Garab G, Hernádi K, 437 Nagy L. 2015. Structural and functional hier- 438 archy in photosynthetic energy conversion— 439 from molecules to nanostructures. Nanoscale 440
Research Letters 10:458-70. 441
Szekeres GP, Nemeth K, Kinka A, Magyar M, Reti B. 403 2015. Controlled nitrogen doping and carboxyl 504 functionalization of multi-walled carbon nano- 505 tubes. Phys. Status Solidi B 252(11):2472-8. 505 Wacker L, Christl M, Synal HA. 2010. Bats: A new Q 15IKÍ tool for AMS data reduction. Nuclear Instruments 547 and Methods in Physics Research B 268:976-79. 549 Wilson BC, Tuchin VV, Tanev S. 2005. Advances in 549
Biophotonics. NATO Science Series: Life & 511
Behavioural Sciences. 511
14 T Szabo et al.
Wolf EL, editor. 2004. Nanophysics and Nano
technology: An Introduction to Modern Concepts in Nanoscience. Weinheim: Wiley-VCH.
Wong MH, Giraldo JP, Kwak S-Y, Koman VB, Sinclair R, Lew TTS, Gili Bisker, Pingwei Liu P, Strano MS. 2017. Nitroaromatic detection and infrared communication from wild-type plants using plant nanobionics. Nature Materials 16:264—72.
Yadav RM, Shripathi T, Srivastava A, Srivastava ON. 2005. Effect of ferrocene concentration on the synthesis of bamboo-shaped carbon-nitrogen
nanotube bundles. J Nanosci Nanotechnol 5(5): 515
820-4. 524
Yang Z, Dai Y, Wang S, Chenga H, Yu J. 2015. In 516 situ incorporation of a S, N doped carbon/sulfur 517 composite for lithium sulfur batteries. RSC 528
Advances 5:78017-25. 518
Yedra L, Eswara S, Dowsett D, Wirtz T. 2016. In-situ 5J0 isotopic analysis at nanoscale using parallel ion 521 electron spectrometry: a powerful new paradigm 522 for correlative microscopy. Scientific Reports 522
6:28705. 524
535