ContentslistsavailableatSciVerseScienceDirect
Peptides
jo u r n al h om ep ag e :w w w . e l s e v i e r . c o m / l o c a t e / p e p t i d e s
Short communication
Effect of PACAP treatment on kidney morphology and cytokine expression in rat diabetic nephropathy
E. Banki
a, P. Degrell
b, P. Kiss
a, K. Kovacs
c, A. Kemeny
d, K. Csanaky
a, A. Duh
a, D. Nagy
d, G. Toth
e, A. Tamas
a, D. Reglodi
a,∗aDepartmentofAnatomyPTE-MTA“Lendulet”PACAPResearchTeam,UniversityofPecs,Pecs,Hungary
bInternalMedicine2/NephrologyCenter,UniversityofPecs,Pecs,Hungary
cDepartmentofBiochemistryandMedicalChemistry,UniversityofPecs,Pecs,Hungary
dDepartmentofPharmacologyandPharmacotherapy,UniversityofPecs,Pecs,Hungary
eDepartmentofMedicalChemistry,UniversityofSzeged,Szeged,Hungary
a r t i c l e i n f o
Articlehistory:
Received2January2013
Receivedinrevisedform4February2013 Accepted4February2013
Availableonline13February2013
a b s t r a c t
Pituitaryadenylatecyclaseactivatingpolypeptide(PACAP)isaneuropeptide,exertingdiverseeffects.One ofitsfrequentlyexaminedfunctionsiscellprotection,whichisachievedmainlyviainhibitingapoptotic, inflammatoryandoxidativeprocesses.Allitsthreereceptors(PAC1,VPAC1,VPAC2)areexpressedinthe kidneyandPACAPhasbeenshowntohaveprotectiveeffectsagainstdifferentrenalpathologies.Diabetic nephropathyistheleadingcauseofendstagerenaldisease.Theaimofthepresentstudywastoinvestigate thepossibleameliorativeeffectofPACAPinstreptozotocin-induceddiabeticnephropathyandtoevalu- ateitsanti-inflammatoryeffectinthismodel.Diabeteswasinducedbyasingleintravenousinjectionof streptozotocin(65mg/kg)inmaleWistarrats.PACAP-treatedanimalswereadministeredip.20gPACAP everysecondday,whileuntreatedanimalsweregivenvehicle.Kidneyswereremovedafter8-weeks survival.Besidesthecomplexhistologicalanalysis(glomerularPASpositivearea/glomerulusarea,tubu- lardamage,arteriolarhyalinosis),expressionofseveralcytokineswasevaluatedbycytokinearrayand Luminexassay.Histologicalanalysisrevealedseverediabeticchangesinkidneysofcontroldiabeticani- mals(glomerularPAS-positiveareaexpansion,tubulardamage,Armanni-Ebsteinphenomenon).PACAP treatmentsignificantlydiminishedthedamage.Diabetickidneysshowedsignificantcytokineactivation comparedtotheirhealthycontrols.PACAPwaseffectiveindownregulationofseveralcytokinesincluding CINC-1,TIMP-1,LIX,MIG,s-ICAM.Toconclude,PACAPiseffectiveinamelioratingdiabeticnephropathy atleastpartlythroughitswell-knownanti-inflammatoryeffect.Theseresultsraisetheopportunityfor theuseofPACAPasapossibletherapeuticorpreventivemethodintreatingthecomplicationsofdiabetes.
©2013ElsevierInc.Allrightsreserved.
1. Introduction
Pituitaryadenylatecyclaseactivatingpolypeptide(PACAP)isa neurotrophicandneuroprotectiveneuropeptide,whichexistsin twoformswith38and27aminoacidresidues:PACAP-38and- 27,respectively[42].PACAPactsthroughthreeGprotein-coupled receptors:thespecificPAC1andthenon-specificVPAC1andVPAC2 receptors.Highestlevelsof PACAPoccurinthecentralnervous systemandendocrineglands[42].However,lowerexpressionhas alsobeenshowninthegastrointestinal,respiratory,cardiovascular andurogenitalsystems[13,42].Basedonthewidespreaddistribu- tion,PACAPhasbeenfoundtohavediverseeffectsinthenervous
∗Correspondingauthorat:DepartmentofAnatomy,UniversityofPecs,7624Pecs, Szigetiu12,Hungary.Tel.:+3672536001x35398;fax:+3672536393.
E-mailaddress:dora.reglodi@aok.pte.hu(D.Reglodi).
systemandperipheralorgans.Oneoftheearlydescribedeffects ofPACAPisitsstrongneuroprotectivepotential.Severalstudies haveproventhatPACAPisageneralcytoprotectivepeptide,exert- ingcell-survivalpromotingeffectsinnumeroustissuesandcells, including endothelialcells, lymphocytes, liver,lungs and ovary [11,28,45,47].
Actionsof PACAP in thekidney are less known.PACAP and itsreceptors(PAC1,VPAC1 andVPAC2)areallexpressedin the human, rat and chicken renal tissues [27]. Some effects have alsobeendescribedinthekidney,suchasvasodilationandrenin secretion[27].However,alargebodyofexperimentaldatasup- portstheprotectiveeffectsofPACAPinthekidney.Itiseffective againsthydrogenperoxide-inducedoxidativestressinvitro[15].In vivo,PACAPisprotectiveagainstischemia/reperfusioninjuryand improvesthesurvivalrateofratsundergonerenalvesselclamping [17,38].PACAPisalsorenoprotectiveagainstmyelomanephropa- thy,subsequentlyevenconfirmedinasinglepatientstudy[2,20].
0196-9781/$–seefrontmatter©2013ElsevierInc.Allrightsreserved.
http://dx.doi.org/10.1016/j.peptides.2013.02.002
SimilarnephroprotectiveeffectcanbeobservedincyclosporineA andgentamicin-inducedinjuries[2,18,20,27].
Based on thesedata, the nephroprotectiveeffects of PACAP arewell-established.However,littleisknownabouttherelation- shipbetweenPACAPandoneofthemostcommonnephropathies, thediabeticnephropathy.Amongalltheclassicaldiabeticcompli- cations(retino-,nephro-andneuropathy),diabeticnephropathy causesthehighestincreaseinmorbidityandmortality.Diabetic nephropathy affects approximately 30–40% of all the diabetic patientsanditistheleadingcauseofchronickidneydiseaseand endstagerenalfailure.Patientssufferingfromdiabeticnephropa- thyshow15–20-foldhighermortalityratecomparedtohealthy subjects.Thediseaseischaracterizedbybothfunctionalandstruc- turalchangesofthekidney.Glomerularhypertrophy,glomerular basement membrane thickening, apoptosis and desquamation of the podocytes are already present in the early stage of the disease. Mesangial matrix expansion, diffuse and nodular glomerulosclerosis,tubulointerstitialfibrosis,tubularatrophyand glycogen-accumulationalsodevelopwiththeprogressionofthe disease(Armanni-Ebsteinphenomenon)[40,48].Inrats,diabetic nephropathycanbemimickedbystreptozotocin-inducedpancre- aticbetacellloss[41].Anearlierstudyhasdescribedprotective effectsofPACAPinearlydiabeticnephropathy[20]andwehave recentlyproventhatPACAPisprotectiveinanothercommoncom- plicationofdiabetes,thediabeticretinopathy[36].Theaimofthe presentstudywastoinvestigatetheprotectivepotentialofinvivo PACAPtreatmentin8weekdiabetes-inducednephropathyandto studytheinvolvementof anti-inflammatorypathwaysbyusing cytokinemeasurements.
2. Materialsandmethods 2.1. Animals
AdultmaleWistarrats(n=19)weighing250–300gwerehoused underlight/darkcyclesof12:12handreceivednormalratchow and drinkingwater ad libitum. Rats were randomlydivided in 4 groups: (1) untreated control (saline i.v. and i.p., n=4); (2) PACAP-treatedcontrol(salinei.v.and20gPACAP1-38i.p.every second day, n=4); (3) untreated diabetic (65mg/kg streptozo- tocin(Sigma,Hungary)i.v.andsalinei.p.,n=6);(4)PACAP-treated diabetic(65mg/kg streptozotocin i.v.and 20g PACAP1-38i.p.
every second day, n=5). Rats were weighed and their blood glucose concentration was measured weekly by blood glucose monitor(Accu-CheckActive,Roche,Hungary).Animalswerecon- sidereddiabeticincasestheyshowedelevatedbloodglucoselevels (>11mmol/l) and theclassical symptoms of diabetes including polyuria,polydipsia,polyphagiaandexcessiveweightloss.After 8weeks,animalsweresacrificedwithanoverdoseofanesthetics andkidneyswereremovedandweighed.Experimentalprocedures werecarriedoutinaccordancewithapprovedprotocols(University ofPecs;BA02/2000-15024/2011).
2.2. Renalhistologyandmorphometricanalysis
Kidneyswerefixedin10%formalin,embeddedinparaffinand 5mthinsectionswerecutwithmicrotome.Sectionswerestained withperiodicacid-Schiffbase(PAS)or withhematoxylin–eosin anddigitalphotosweretaken.DiastasedigestedPASreactionafter absolutealcoholfixationwasalsoperformedtoprovetheglycogen contentof tubulargranules.Renalhistologicalanalysiswascar- riedoutbyanephropathologistblindedtothetreatmentgroups (PD).Atleast10glomerulioneachslidewereexaminedfordeter- minationofglomerularchanges[40].Thedegreeofaccumulated glycogengranules(Armanni-Ebsteinphenomenon)wasevaluated
asanindicatoroftubulointerstitiallesions.Analysiswascarriedout at400×magnification.MagentastainingofintraglomerularPAS positiveareashowsthemesangialmatrixand basalmembrane, whiletubularPASpositivegranules,Armanni-Ebsteinlesionsare thesignoftubulardamage.AdobePhotoshopprogramwasused tomarktheglomerulusmanually,thenitwasconvertedtoblack, whiletheremainingareatowhite.Thepropermagentaofintra- glomerularPASpositiveareaandtubularglycogengranuleswere alsomarkedandconvertedtoblackwiththesamemethod.Finally in every picture the “area of interest”was converted to black withtheremainingareabecomingwhite,thuscreatingblackand white pictures. Black areas in the vision fields were measured usingScionImageprogram.Finally,theintraglomerularPASpos- itiveareawasexpressedagainsttheareaofthesameglomerulus, whileglycogengranulesagainstthevisionfield.Arteriolarhyali- nosis,alackofsmoothmuscleinthearteriolarwall,wasevaluated onhematoxylin-eosinsectionsaccordingtothefollowingscore:“0”
arteriolewithoutanysignofhyalinosis,“1”,“2”,“3”,“4”withhyali- nosisaffecting¼,½,
¾
ortheentirecrosssectionofthearteriole, respectively.2.3. Cytokinemeasurements
Thesemiquantitativecytokinearraywasperformedfromkid- ney homogenates using Rat Cytokine Array kit (R&D Systems;
BiomedicaHungaria,Hungary).Thesearraysarebasedonbinding between sample proteinsand carefully selected captured anti- bodiesspottedonnitrocellulosemembranes.Weexaminedtissue samples fromall the 4 groups:control, PACAP-treated control, diabeticandPACAP-treateddiabeticgroups.Thearraywasper- formedasdescribedbythemanufacturer,similarlytoourprevious study[25].Briefly,kidneysampleswerehomogenizedinPBSwith proteaseinhibitor.TritonX-100wasaddedtothefinal concen- trationsof 1%.Afterblocking thearraymembranes for 1hand addingthereconstituteddetectionantibodycocktailforanother 1hat roomtemperature, themembranes wereincubated with 1ml of tissue homogenates at 2–8◦C overnight on a rocking platform. After washing with buffer three times and addition ofhorseradishperoxidase-conjugatedstreptavidintoeachmem- brane,weexposedthemtoachemiluminescentdetectionreagent (AmershamBiosciences,Hungary),thensideuptoanX-rayfilm cassette.Thearraywasrunin duplicatesets.Resultsarerepre- sentedinpixel densityand arecomparedin asemiquantitative fashion.
LuminexMultiplexImmunoassaywasperformedasdescribed previously[25].Briefly,soluble intercellularadhesionmolecule- 1 (sICAM1) and L-selectin levels were determinated using customizedFlurokineMAPRatBaseKit(R&DSystems).Theexper- imentwasperformedaccordingtothemanufacturer’sinstructions (R&DSystems).Followingpreviousoptimizations,allsampleswere testedundilutedinablind-fashion.Luminex100devicewasused fortheimmunoassayandLuminex100ISsoftwarefortheanalysis ofbeadmedianfluorescenceintensity.Sampleswerehomogenized withRPMI-1640(GIBCO)containing1%proteaseinhibitorcock- tail,sampleswereusedin20mg/mlconcentrations.Allthetests wereruninduplicate.50lvolumeofeach sample,control,or standardwasaddedtoa96-wellplate(providedwiththekit)con- taining 50lofantibody-coated fluorescentbeads.Biotinylated secondaryandstreptavidin-PEantibodieswereaddedtotheplate withalternateincubationandwashingsteps.Afterthelastwash- ingstep,100lofthebufferwasadded tothewells;theplate wasincubatedand readontheLuminex100arrayreader,using afour-PLregressioncurvetoplotthestandardcurve.Datawere subsequentlyanalyzedusingtheLuminex100managersoftware.
ResultsaregiveninpgL-selectinors-ICAM/gwettissue.
Fig.1.Histologicalanalysisofcontrolanddiabetickidneys.Representativehistologicalsectionsofglomeruli(rowA)andrenaltubules(B).Sectionsofcontrol(AandB), PACAP-treatedcontrol(AandB),diabetic(AandB),PACAP-treateddiabetic(AandB)animals.ControlandPACAP-treatedcontrolanimalsshowednormalglomerular andtubularstructure.DiabetickidneysshowedsevereintraglomerularPASpositiveareaexpansionandglomerulardamage(A)andseveretubularatrophyanddeposition ofglycogengranules,calledArmanni-Ebsteinphenomenon(B,arrows).PACAP-treatmentwaspreventiveagainstdiabeticchanges,milderdamagewasobservedinthese sections(AandB).Periodicacid-Schiff(PAS)reaction.Calibrationmark:20m.Representativehistologicalsectionsofarterioleswithaseverityscoreof0,1,2,3,4 (C,C,C,C,Crespectively).Hematoxylin–eosinstaining.Calibrationmark:20m.PASpositiveareaperglomerulararearatio(D)***p<0.0001vs.allothergroups (control,control+PACAP,diabetes+PACAPgroups).Glycogengranules(Armanni-Ebsteinphenomenon)pervisionfieldratio(E)***p<0.0001vs.allothergroups(control, control+PACAP,diabetes+PACAPgroups).Vascularhyalinosisscore(F).Significanthyalinosiswasobservedindiabeticanimals,whileinthePACAP-treateddiabeticanimals therewerenosignsofthePASpositivearteriolarwallthickening.***p<0.0001diabetesvsallothergroups(control,control+PACAP,diabetes+PACAPgroups).
2.4. Statisticalanalysis
Statistical analysis wasperformed by Microsoft Office Excel andGraphPadsoftware.Repeatedmeasuresanalysisofvariance (ANOVA)withBonferronicorrectionwasusedtodetectsignificant differencesbetweengroups.p-Valuelessthan0.05wasconsidered tobestatisticallysignificant.
3. Results
3.1. Bodyweight,totalkidneyweighttobodyweightratioand bloodglucoselevels
Thebodyweightof theanimalsafter8 weeksoftreatments was365±29gintheintactcontrolsand351±31ginthePACAP- treatedgroup.Diabeticanimalsshowedasignificantdecreasein theirweight:wemeasured230±17ginthediabeticand260±19g inthePACAP-treateddiabeticgroup.However,theslightdifference betweendiabeticandPACAP-treateddiabeticratsdidnotresultto bestatisticallysignificant.PACAPtreatmentalonedidnotresultin significantchangeinkidney/bodyweight(6.44±0.13)compared totheuntreatedcontrolkidneys(6.68±0.09).Indiabeticanimals, wecouldobserveasignificantincreaseinthisratio(13.80±1.36), similarlytoobservationsbyothers[6].PACAPtreatmentindia- beticanimalsslightlydecreasedthisratio,however,thiswasnot statisticallysignificant(12.33±0.37).Therewasnodifferencein
the weekly blood sugar level in the intactcontrol and PACAP- treatedgroupsorbetweenthediabeticcontrolandPACAP-treated groups.Attheendoftheexperiment(atweek8)thebloodglucose levelwas7.85±0.42and7.55±0.36mmol/Lintheintactcontrol andPACAP-treatedintactanimals,respectively.Inthediabeticani- mals,thesevalues were30.13±1.75mmol/Linthediabeticrats and32.42±0.69mmol/LinthePACAP-treateddiabeticanimals.
3.2. PACAPattenuateddiabeticnephropathy:renalhistologyand morphometricanalysis
In control animals, normal glomerular structure was found withoutanytubularor glomerularinjury and PACAPtreatment for8weeksdidnotcauseanychanges.Kidneysfromdiabeticani- malsshowedsignsofseverenephropathy,withmesangialmatrix expansionandabundantglycogengranules(Fig.1A–D).Ahallmark of diabeticchangesindicating theprogression ofthemesangial changes indiabeticnephropathy is theratioof intraglomerular PASpositiveareaperglomerulararea.Thissignificantlyincreased in the diabetic rats, while PACAP treatment could effectively counteract this lesion: PACAP-treated diabetic animals showed no significant differencecompared with intact control kidneys (Fig.1AandD).Anotherveryimportantcharacteristicsofdiabetic nephropathyistheappearanceofglycogendepositsinthetubules, alsocalledArmanni-Ebsteinphenomenon[48].Thisfeaturewas completelymissingfromcontrolkidneys,whilethepresenceof
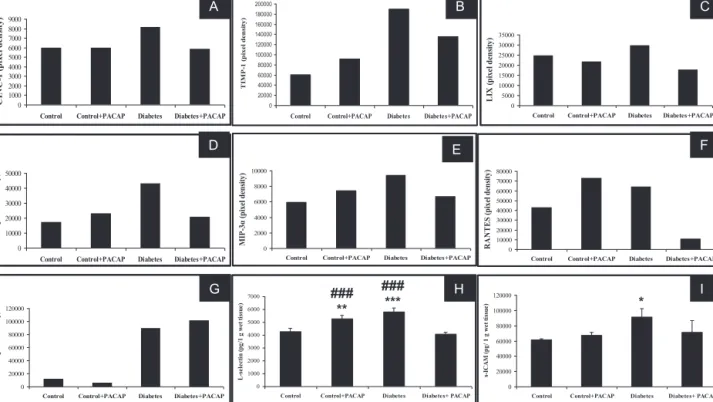
Fig.2.DataofthesemiquantitativecytokinearrayofCINC-1,TIMP-1,LIX,MIG,RANTES,MIP-3␣,CNTF(A)–(G).Dataarepresentedasmeanpixeldensity.Measurement ofL-selectinands-ICAMbyLuminexassay(H)and(I).Dataarepresentedasmeanpg/gwettissue±SEM.*p<0.05,**p<0.001,***p<0.0001vs.control,###p<0.0001vs.
diabetes+PACAP.
glycogendepositsmarkedlyincreasedindiabeticrats.Incontrast, theamountofglycogen depositswassignificantlylower inthe PACAP-treateddiabeticgroupthanintheuntreateddiabeticgroup (Fig.1BandE).Acharacteristicfeatureofmicroangiopathyisvas- cularhyalinosis,whichissparinglypresentinthecontrolgroups.
Thedegreeofarteriolarhyalinosiswassignificantlyhigherinthe diabeticgroupcomparedtocontrolanimals,whilePACAPtreat- mentwasabletocompletelypreventthedevelopmentofhyalinosis (Fig.1CandF).
3.3. Cytokinemeasurements
PACAP treatment had no effect on the level of numerous cytokines in control animals, however, it elevated the expres- sionof afew cytokines: tissue inhibitorof metalloproteinase 1 (TIMP-1),monokineinducedbygammainterferon(MIG/CXCL9), macrophage inflammatory protein 3␣ (MIP-3␣), regulated and normalTcellexpressedandsecreted(RANTES,CCL5),L-selectin (CD62L/LECAM-1)anddecreasedlipopolysaccharide-inducedCXC chemokine(LIX/CXCL5)andciliaryneurotrophicfactor(CNTF)lev- els.Diabetesstronglyincreasedtheexpressionofcytokineinduced neutrophilchemokine(CINC-1),TIMP-1,LIX,MIG,MIP-3␣,RANTES, CNTF and L-selectin. Treatment with 20g PACAP markedly decreasedtheabove-mentionedcytokinesandchemokines,with someofthemreachingcontrollevels(Fig.2A–I).Nochangeswere observedinotherspots:Fractalkine,thymuschemokineandinter- leukins(datanotshown).
4. Discussion
In the present study we showed that in vivo PACAP treat- mentisprotectiveindiabeticnephropathyandthisameliorative effect is at least partly due to the anti-inflammatory effect of PACAP.Ourobservationscompletethelistofkidneypathologies againstwhichPACAP hasprotective effects.Among others,it is
effectiveagainstoxidativestress,gentamicin-inducednephropa- thy, myeloma and ischemia-induced injuries and cyclosporine A-inducedlesion[2,15,18,20,38].Anearlierstudyhasevenmen- tionedthatPACAPhasprotectiveeffectsinearly(2weeks)diabetic nephropathy in rats [20]. Our present results, with prolonged survival time, are thus in accordance with these observations and complete these findings withdetailed histologicalanalysis andcytokinemeasurements.Furthermore,weappliedlong-term PACAPadministrationforthefirsttimeinthetreatmentofrenal diseases,provingthatPACAPtreatmentcanbeeffectiveevenin chronictherapy.
OurstudyshowedthatPACAPmarkedlyattenuatedthecharac- teristicpathologicalalterationsobservedindiabeticratswithout alteringtheblood glucoselevels.IncreasedintraglomerularPAS positiveareaisareliableindicatorofbasementmembranethick- ening and glomerular mesangial matrix expansion, which are characteristicfordiabeticglomerulopathy[40].Inthepresentstudy wefoundsignificantexpansionofglomerular PAS-positive area caused byhyperglycemia aftera singlestreptozotocin injection similarlytoothers[22].Hyperglycemiastimulatesthepatholog- icalaccumulationoftheotherwise physiologicalcomponentsin the extracellular matrix, like laminin, fibronectin, collagene IV throughoverexpressionofTGF-1[12].Changesinthetubuloint- erstitiummaypossessevenhigherimportanceintheprogression ofdiabetickidneydisease[39].Tubularvacuolationonautopsyis consideredtobepathognomictodeathduetodiabeticcoma.In ourstudy,tubulardamagewasevaluatedbasedontheamountof glycogengranuleswithintheproximaltubules,calledArmanni- Ebsteinphenomenon[19,48].Arteriolarhyalinosisoccursinboth afferentand efferentarteriolesin thediabetickidney,however, efferentarteriolarhyalinosisisconsideredtobecharacteristicfor diabeticnephropathy[35].KidneysofPACAP-treateddiabeticani- malsshowedsignificantlymilderglomerular,tubularandvascular changes.
Asfarastheprotectivemechanismisconcerned,weinvesti- gatedtheanti-inflammatoryeffectsofPACAPinthepresentstudy.
PACAPiswell-knownforitsanti-inflammatoryactionsalsoinsome oftheabove-mentionedrenalpathologicalconditions[27].Recent studiesindicatethatinflammationisoneofthekeymechanisms inthedevelopmentofdiabeticnephropathyinadditiontohyper- glycemia and hemodynamicfactors [24]. Mostrenal cells, such asendothelial,mesangial,epithelialcellsandpodocytesbecome activated and produce proinflammatory cytokines, chemokines and adhesion molecules upon injury. These chemoattractant moleculesinduceinfiltrationofseveralimmunecells,likemono- cytes, neutrophils and lymphocytes intothe kidney, leadingto tubulointerstitialfibrosis,glomerulosclerosis,tubularatrophyand vasculardamage[1,46].PACAPhasbeenfoundtoaltercytokine expressioninnumerousstudies.Wehavedescribed earlierthat PACAPcounteractedseveralchangesincytokineandchemokine expressioninducedbyintestinalandkidneyischemia/reperfusion andretinalischemia[15,25,37].Inthepresentstudywefoundthat PACAPreducedthediabetes-inducedelevationofCINC-1,TIMP-1, LIX,MIG,MIP-3␣,RANTES,L-selectinands-ICAM.Allthesefactors playapotentialroleindiabeticnephropathy.Forexample,CINC-1, oneofthemajorneutrophilchemoattractantsstimulatedbyoxida- tivestress and ischemia/reperfusion, isalso present in diabetic nephropathy[7,23].LevelsofTIMP-1,LIX,MIGands-ICAM,arealso elevatedintheurineandserumofdiabeticnephropathypatients [4,5,14,16].MIP-3␣mRNA and proteinwere foundtobe upre- gulatedbyhyperglycemiabothinvivoandinvitroandelevated urinaryMIP-3␣aftertransplantationmightcontributetoallograft rejection[26,43].TheeffectsofPACAPontheinjury-inducedupre- gulatedexpressionofinflammatorycytokinesseemtobeamore generalphenomenon,sincewefoundsimilareffectsintheretina andintestinaltissue[25,37].
We found an interesting pattern of RANTES and L-selectin expression in our treated groups. PACAP alone increased the expression of both, while it descreased theirlevels in diabetic animals.TheeffectofPACAPonsomecytokinesandchemokines iscontradictoryandbothstimulatingandinhibitingeffectshave beendescribeddependingonthecelltypeinjury [9,37]. Earlier studieshaveshownupregulatedRANTESindiabetickidneys,cor- relating withproteinuriaand expressionofNFB, suggestiveof progressivediabeticnephropathy.Earlierstudieshavefoundthat PACAPcaninducethesecretionofRANTESinastrocytesandcortical neurons[3,31],mostprobablyassociatedwithincreasedneuro- protection.However,inotherstudies,PACAPreducedtheelevated expressionofRANTESintheretinaandinmicroglialcells[9,37].
The exact role of PACAP in renal RANTES expression needsto befurtherinvestigated,butthefindingthatPACAPcouldreduce thediabetes-inducedelevationsuggestsanephroprotectiveaction.
Arapid increasein L-selectin,a celladhesionmoleculerespon- siblefor therenalmononuclear infiltration,hasbeendescribed afterrenalischemia/reperfusioninjury[10,32,34].Thefindingthat PACAPreducedthediabetes-inducedelevationofL-selectinsug- gestsaprotectiveeffect,buttheinitialstimulatoryeffectofPACAP incontrolanimalsneedstobefurtherclarified.Incontrasttoother cytokines,PACAPstimulatedtheexpressionofthetrophicfactor CNTFindiabetickidneys,indicating anotherpossibleprotective mechanismsimilarlytoearlierdescriptions[29].
Insummary,ourpresentstudyshowedtheprotectiveeffects ofinvivoPACAPtreatmentindiabeticnephropathybyinvolving anti-inflammatoryactions.Ourstudyfurthersupportstheimplica- tionthatPACAPmighthaveprotectivepotentialindiabetesandits complications.PACAPisabletoincreasepancreaticbetacellpro- liferation,enhanceinsulinsecretionandithasbeenshowntobe protectiveintwomajordiabeticcomplications:retinopathyand nephropathy[30,33,36,44].Thus, PACAPisagood candidatefor therapeuticinterventionsindiabetesatmultiplelevels.Oneofthe drawbacksofsystemicPACAPtreatmentistherapiddegradation ofthepeptideinthebloodbydipeptidylpeptidase-IV[DPP-IV,42].
Inhibitorsofthisenzymearebeingusedinthetreatmentofdia- beticpatientsfortheincreasedlevelofglucagon-likepeptide-1[8].
Protectiveeffectshavebeendescribedevenindiabeticnephropa- thy[21].Arecentpaperhassuggestedthatthetherapeuticeffects ofDPP-IVinhibitorscouldinvolvesofarunknownmechanisms, suchasincreasedPACAPlevels[8].Whethertheprolongedpres- enceofPACAPinthecirculationisprotectiveindiabetes-related complications,istobedeterminedbyfutureinvestigation.
Acknowledgements
This study was supported by OTKA K104984, TAMOP (4.2.1.B-10/2/KONV-2010-002, 4.2.2.B-10/1-2010-0029, 4.2.2.A- 11/1/KONV-2012-0024),ArimuraFoundation,PTE-MTA“Lendület”
Program.
References
[1]AbbateM,ZojaC,RemuzziG.Howdoesproteinuriacauseprogressiverenal damage?JAmSocNephrol2006;17:2974–84.
[2] ArimuraA,LiM,BatumanV.Potentialprotectiveactionofpituitaryadenylate cyclase-activatingpolypeptide(PACAP38)oninvitroandinvivomodelsof myelomakidneyinjury.Blood2006;107:661–8.
[3]BrennemanDE,HauserJM,SpongC,PhillipsTM.Chemokinereleaseisasso- ciatedwiththeprotectiveactionofPACAP-38againstHIVenvelopeprotein neurotoxicity.Neuropeptides2002;36:271–80.
[4]BrunoCM,ValentiM,BertinoG,ArdiriA,BrunoF,CunsoloM,etal.Plasma ICAM-1andVCAM-1 levelsintype2diabeticpatients withandwithout microalbuminuria.MinervaMed2008;99:1–5.
[5] CataniaJM,ChenG,ParrishAR.Roleofmetalloproteinasesinrenalpathophys- iologies.AmJPhysiolRenalPhysiol2007;292:F905–11.
[6]ChenS,HongSW,Iglesias-delaCruzMC,IsonoM,CasarettoA,ZiyadehFN.The keyroleofthetransforminggrowthfactor-betasysteminthepathogenesisof diabeticnephropathy.RenFail2001;23:471–81.
[7]CuginiD,AzzolliniN,GagliardiniE,CassisP,BertiniR,ColottaF,etal.Inhibition ofthechemokinereceptorCXCR2preventskidneygraftfunctiondeterioration duetoischemia/reperfusion.KidneyInt2005;67:1753–61.
[8] DarsaliaV,OrtsaterH,OlverlingA,DarlofE,WolbertP,NystromT,etal.The DPP-4inhibitorlinagliptincounteractsstrokeinthenormalanddiabeticmouse brain.Diabetes,PMID23209191,inpress.
[9] DelgadoM,JonakaitGM,GaneaD.Vasoactiveintestinalpeptideandpituitary adenylatecyclase-activatingpolypeptideinhibitchemokineproductioninacti- vatedmicroglia.Glia2002;39:148–61.
[10]DragunD, HoffU, ParkJK,QunY,SchneiderW,LuftFC,etal.Ischemia- reperfusioninjuryinrenaltransplantationisindependentoftheimmunologic background.KidneyInt2000;58:2166–77.
[11]ElekesK,SandorK,MoriczA,KereskaiL,KemenyA,SzokeE,etal.Pitu- itaryadenylatecyclaseactivatingpolypeptideplaysananti-inflammatoryrole inendotoxin-inducedairwayinflammation:invivostudywithgene-deleted mice.Peptides2011;32:1439–46.
[12]Falk RJ, Scheinman JI, Mauer SM, Michael AF. Polyantigenic expansion of basement membrane constituents in diabetic nephropathy. Diabetes 1983;32(Suppl.2):34–9.
[13]GirardBM,MalleySE,BraasKM,MayV,VizzardMA.PACAP/VIPandreceptor characterizationinmicturitionpathwaysinmicewithoverexpressionofNGF inurothelium.JMolNeurosci2010;42:378–89.
[14]HigurashiM,OhyaY,JohK,MuraguchiM,NishimuraM,TerawakiH,etal.
IncreasedurinarylevelsofCXCL5,CXCL8andCXCL9inpatientswithType2 diabeticnephropathy.JDiabetesComplications2009;23:178–84.
[15]HorvathG,BrubelR,KovacsK,ReglodiD,OpperB,FerenczA,etal.Effectsof PACAPonoxidativestress-inducedcelldeathinratkidneyandhumanhepa- tocytecells.JMolNeurosci2011;43:67–75.
[16]KanauchiM,NishiokaH,NakashimaY,HashimotoT,DohiK.Roleoftissue inhibitorsofmetalloproteinaseindiabeticnephropathy.NipponJinzoGakkai Shi1996;38:124–8.
[17]KhanAM,LiM,Abdulnour-NakhoulS,MaderdrutJL,SimonEE,BatumanV.
Delayedadministrationofpituitaryadenylatecyclase-activatingpolypeptide 38amelioratesrenalischemia/reperfusioninjuryinmicebymodulatingToll- likereceptors.Peptides2012;38:395–403.
[18] KhanAM,LiM,BrantE,MaderdrutJL,MajidDS,SimonEE,etal.Renoprotec- tionwithpituitaryadenylatecyclase-activatingpolypeptideincyclosporine A-inducednephrotoxicity.JInvestigMed2011;59:793–802.
[19]KockKF,VestergaardV.Armanni-Ebsteinlesionsofthekidney:diagnosticof deathindiabeticcoma.ForensicSciInt1994;67:169–74.
[20]LiM,MaderdrutJL,LertoraJJ,ArimuraA,BatumanV.Renoprotectionbypitu- itaryadenylatecyclaseactivatingpolypeptideinmultiplemyelomaandother kidneydiseases.RegulPept2008;145:24–32.
[21] LiuWJ,XieSH,LiuYN,KimW,JinHY,ParkSK,etal.DipeptidylpeptidaseIV inhibitorattenuateskidneyinstreptozotocin-induceddiabeticrats.JPharma- colExpTher2012;340:248–55.
[22]Matavelli LC, Siragy HM. Reduction of aldosterone production improves renaloxidativestressandfibrosisindiabeticrats.JCardiovascPharmacol 2013;61:17–22.
[23]MizutaniA,OkajimaK,UchibaM,NoguchiT.ActivatedproteinCreduces ischaemia/reperfusion-inducedrenalinjuryinratsbyinhibitingleukocyteacti- vation.Blood2000;95:3781–7.
[24]Navarro-GonzálezJF,Mora-FernándezC,MurosdeFuentesM,García-Pérez J. Inflammatorymolecules and pathways inthepathogenesis ofdiabetic nephropathy.NatRevNephrol2011;7:327–40.
[25] NedvigK,WeberGyNemethJ,KovacsK,ReglodiD,KemenyA,etal.Intestinal autotransplantationusingPACAP-38-containingpreservationsolution.JMol Neurosci2012;48:788–94.
[26]QiW,ChenX,ZhangY,HolianJ,MreichE,GilbertRE,etal.Highglucose inducesmacrophageinflammatoryprotein-3alphainrenalproximaltubule cellsviaatransforminggrowthfactor-beta1dependentmechanism.Nephrol DialTransplant2007;22:3147–53.
[27]ReglodiD,KissP,HorvathG,LubicsA,LaszloE,TamasA,etal.Effectsofpituitary adenylatecyclaseactivatingpolypeptideintheurinarysystem,withspecial emphasisonitsprotectiveeffectsinthekidney.Neuropeptides2012;46:61–70.
[28] ReglodiD,KissP,SzabadfiK,AtlaszT,GabrielR,HorvathG,etal.PACAPis anendogenousprotectivefactor– insightsfromPACAPdeficientmice.JMol Neurosci2012;48:482–92.
[29]RezendeLF,SantosGJ,CarneiroEM,BodscheroAC.Ciliaryneurotrophicfactor protectsmiceagainststreptozotocin-inducedtypeIdiabetesthroughSOCS3:
theroleofSTAT1/STAT3ratioin-celldeath.JBiolChem2012;287:41628–39.
[30]SakumaY, RicordiC,MikiA,YamamotoT,MitaA,BarkerS,etal.Effect ofpituitaryadenylatecyclase-activatingpolypeptideinislettransplantation.
TransplantProc2009;41:343–5.
[31]SanchezA,TripathyD,GrammasP.RANTESreleasecontributestothepro- tectiveactionofPACAP38againstsodiumnitroprussideincorticalneurons.
Neuropeptides2009;43:315–20.
[32] ShikataK,SuzukiY,WadaJ,HirataK,MatsudaM,KawashimaH,etal.L-selectin anditsligandsmediateinfiltrationofmononuclearcellsintokidneyintersti- tiumafteruretericobstruction.JPathol1999;188:93–9.
[33]Shintani N,TomimotoS,HashimotoH,Kawaguchi C, BabaA. Functional rolesoftheneuropeptidePACAPinbrainandpancreas.LifeSci2003;74:
337–43.
[34]SpringerTA.Trafficsignalsforlymphocyterecirculationandleukocyteemigra- tion:themultistepparadigm.Cell1994;76:301–14.
[35] StoutLC,KumarS,WhortonEB.Insudativelesions-theirpathogenesisand associationwithglomerularobsolescenceindiabetes:adynamichypohesis
basedonsingleviewsofadvancinghumandiabeticnephropathy.HumPathol 1994;25:1213–27.
[36]SzabadfiK,AtlaszT,KissP,ReglodiD,SzaboA,KovacsK,etal.Protective effectsoftheneuropeptidePACAPindiabeticretinopathy.CellTissueRes 2012;348:37–46.
[37]SzaboA,DanyadiB,BognarE,SzabadfiK,FabianE,KissP,etal.EffectofPACAP onMAPkinases,Aktandcytokineexpressionsinratretinalhypoperfusion.
NeurosciLett2012;523:93–8.
[38]Szakaly P,Kiss P,LubicsA,MagyarlakiT,TamasA,RaczB,etal. Effects of PACAP on survival and renal morphology in rats subjected to renal ischemia/reperfusion.JMolNeurosci2008;36:89–96.
[39] TaftJL,NolanCJ,YeungSP,HewitsonTD,MartinFI.Clinicalandhistological correlationsofdeclineinrenalfunctionindiabeticpatientswithproteinuria.
Diabetes1994;43:1046–51.
[40]TervaertTW,MooyaartAL,AmannK,CohenAH,CookHT,DrachenbergCB, etal. Pathologicclassificationofdiabeticnephropathy.JAmSocNephrol 2010;21:556–63.
[41] Tesch GH, Allen TJ. Rodent models of streptozotocin-induced diabetic nephropathy.Nephrology(Carlton)2007;12:261–6.
[42] VaudryD,Falluel-MorelA,BourgaultS,BasilleM,BurelD,WurtzO,etal.Pitu- itaryadenylatecyclase-activatingpolypeptideanditsreceptors:20yearsafter thediscovery.PharmacolRev2009;61:283–357.
[43]WoltmanAM,deFijterJW,vanderKooijSW,JieKE,MassacrierC, Caux C,etal.MIP-3alpha/CCL20inrenaltransplantationanditspossibleinvolve- mentasdendriticcellchemoattractantinallograftrejection.AmJTransplant 2005;5:2114–25.
[44]YamamotoK,HashimotoH,TomimotoS,ShintaniN,MiyazakiJ,TashiroF,etal.
OverexpressionofPACAPintransgenicmousepancreaticbeta-cellsenhances insulinsecretionandamelioratesstreptozotocin-induceddiabetes.Diabetes 2003;52:1155–62.
[45]YuR,GuoX,HuangL,ZengZ,ZhangH.ThenovelpeptidePACAP-TATwith enhanced traversing abilityattenuatestheseverelunginjuryinducedby repeatedsmokeinhalation.Peptides2012;38:142–9.
[46]Zandi-Nejad K, Eddy AA, Glassock RJ, Brenner BM. Why is proteinuria an ominous biomarker of progressive kidney disease. Kidney Int Suppl 2004;92:S76–89.
[47]ZhangH,YuR,LiuX,GuoX,ZengZ.TheexpressionofPAC1increasesin thedegenerativethymusandlowdosePACAPprotectsfemalemicefrom cyclophosphamideinducedthymusatrophy.Peptides2012;38:337–43.
[48] ZhouC,YoolAJ,NolanJ,ByardRW.Armanni-Ebsteinlesions:aneedforclari- fication.JForensicSci2013;58(Suppl1):S94–8.