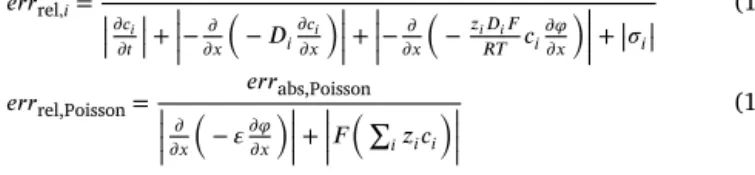Contents lists available atScienceDirect
Heliyon
journal homepage:www.cell.com/heliyon
Research article
Adaptive moving mesh algorithm based on local reaction rate
Viktória Koncz
a,∗, Ferenc Izsák
b, Zoltán Noszticzius
a, Kristóf Kály-Kullai
aaDepartmentofPhysics,BudapestUniversityofTechnologyandEconomics,1521Budapest,Hungary
bDepartmentofAppliedAnalysisandComputationalMathematics,MTAELTENumNetResearchGroup,EötvösUniversity,1518Budapest,Hungary
A R T I C L E I NF O A B S T R A C T
Keywords:
AdaptiveFEM Movingmesh
Reaction-diffusionsystems Acid-basediode COMSOLMultiphysics
Anempiricalmeshadaptionalgorithmisintroducedformodelingone-dimensionalreaction-diffusionsystems withlargemovinggradients.Ournewalgorithmisbasedontherevelation,thatinreaction-diffusionsystems thehighmovingconcentrationgradientsappearnearbytotheregionwheretherateofreactionismaximal, thusthelocalreactionratecanbeusedtocontrolthemeshadaption.Wefound,thatthemainadvantage of sucha method isitssimplicityandeasyimplementation. Asanexamplewestudyanacid-base diode, wherelargemovinggradientsappear.ThemathematicalmodelofthediodecontainsseveralparabolicPDEs, coupledwithoneellipticPDE.An𝑟-refinementtechniqueisusedandattachedtothecommercialfiniteelement solverCOMSOL.Weinvestigatedthetime-dependentsalteffectsofthediodewithourdevelopedalgorithm.
Ourmeshadaptionmethodisadvantageousformodelingofanyreaction-diffusionsystemswithlocalized high concentrationgradients.
1. Introduction
Acid-basediodes,whicharecomplexreaction-diffusion-ionicmigra- tion systems, couldtheoretically be used asion-sensors. They could evenformpartsofso-calledlabonachipelectroanalyticaldevicesbe- causeoftheirsimplicity.Wewouldliketomaterializesuchsensorsas ourfinalaim.Toachievethisaim,itisimportanttostudyandmodel thetimeevolutionoftheeffects,whichwouldbethebasisoftheirap- plicationasionsensor.
Inanelectrolytediodeahydrogelcylinderconnectsanalkalineand anacidicreservoir.This waydiffusion,migrationandreactionofthe componentsareallowed,butadvectionandconvectionarerestrained, unlikeinpurewater.Duetotheappliedelectricpotentialbetweenthe edgesofthegelcylinder,hydrogene(H+)andhydroxil(OH−)ionscan getinside theconnectingelement, wherewateris formedfrom their reaction.
Itmeans,thatsomewhereinthemodeleddomainthereisareaction zone,wherethischemicalacid-basereactionoccurs.Thisreactionzone ischaracterizedbylargeconcentrationandpotentialgradients.Dynam- icalsimulationsoftransientbehaviorinsuchsystemscanbedifficult duetothelargemovinggradientsofthemodelvariables(concentra- tionandpotential).Themodeledreaction-diffusionsystemisdescribed byasetofparabolicpartialdifferentialequations(PDEs,in thiscase
*
Correspondingauthor.E-mailaddress:viki.koncz@gmail.com(V. Koncz).
massbalanceequationsforthechemicalcomponents)andoneelliptic partialdifferentialequation(Poisson-equation).
After the spatial discretization of the domain a system of ordi- narydifferentialequationsisobtained,whichisintegratedforwardby timenumerically(methodof lines).Regardlessthespatialdiscretiza- tionmethod(e.g.finitedifferencemethod,finiteelementmethod)the appliedmeshhasspecialimpactonthenumericalsolvability,speedof convergence,andapproximationerror.Thehigherthenumberofgrid points used,the smallertheerror willbe.However, havingadense gridiscomputationallyexpensive,demandingalargechunkofmemory andanextendedruntime(Farmagaetal., 2011,Zimmerman,2006).
Ifthesolutionshaveregions ofhighspatialactivity,astandarduni- formfixed-gridtechniqueiscomputationallyinefficient,sincetoafford anaccuratenumericalapproximationandreachconvergence,itshould contain,ingeneral,averylargenumberofgridpoints.Themeshneeds tobelocallyrefined.Moreover,iftheregionsofhighspatialactivityare movingintime,likesteepmovingfrontsinreaction-diffusionsystems, thenthemeshshouldbeadaptedaccordingly.
Regardingtothespatialgridadaptionstrategies,atleastthreedif- ferentmethodscanbedistinguished:
• 𝑟-refinementorthemovingmesh(relocation)method:inthiscase fixednumber ofgrid points isused, andthe positionsof points move.
https://doi.org/10.1016/j.heliyon.2020.e05842
Received2October2020;Receivedinrevisedform19November2020;Accepted21December2020
2405-8440/©2020PublishedbyElsevierLtd.ThisisanopenaccessarticleundertheCCBY-NC-NDlicense(http://creativecommons.org/licenses/by-nc-nd/4.0/).
•ℎ-refinementorthelocalrefinementmethod:thenumberofgrid pointsisnotfixed.Pointsareaddedtoorremovedfromthegrid, accordingtothelocalrequirements,theoriginalgridpoints stay inthesamelocation.Practically,thespatialgridstepℎislocally decreasedorincreased.
•𝑝-refinement:inthisapproachafixedmeshisused,andthepoly- nomialdegreeofbasis(𝑝)alters.
Certainly,thecombinationofthesemethodsexists(Schwab,1999).
Although the moving meshmethod has been less popular than the localrefinementmethod,whichisduetothedifficultyofderivingsuit- ablegoverningequationsfortheadaptivemapping,ithassomeuseful featuresinnumericalcomputations.E.g.itis easytoimplement,and coupletoanyfixedmeshsoftware,anddifficultiesfromthechangein numberofgridpointscanbeavoided(Pˇribyletal.,2006).
Usuallythemovingmeshmethodsarebasedontheequidistribution principle(Huangetal.,1994),meaningselectingmeshpointsinsucha way,thatanempiricalerrorisequalizedovereachsubintervalonthe mesh.Theerrordistributionisfollowedbyamonitorfunction,which isderivedfromthefirstandsecondderivativesofdependentvariables.
(Oftenthisdoesnotresembletonumericalerrors.)
Intheliteraturetherearemanyexamplesaboutthesuccessfulnu- mericalmodelingofsimilarPDEsliketheonesdescribingthebehavior ofthediode.Mostofthemethodsrequirethecompletedevelopment andfullimplementationofthespatialdiscretizationandtimeintegra- tionof themodelequations.A movingmeshmethodwas developed (ZegelingandKok,2004) tosolvetheequationsofreaction-diffusion systems, namelytheGray-ScottandBrusselator models.Asystemof adaptivemeshPDEsdescribingthemeshmovementissetupandsolved (inthiscasedecoupledfromtheoriginalPDEsystem),infactthesolu- tionistherealizationofacoordinatetransformationbetweenphysical andcomputationalcoordinates. Theadaptive meshPDEsarederived fromtheminimizationofaso-calledmesh-energyfunctional.Thevari- ationalapproachofthemovingmeshmethodsisgeneral(Caoetal., 2003). Usually the original PDEs are coupled with the mesh PDEs (ZegelingandKeppens,2001).Moreexamplesaboutan𝑟-methodare foundin(Coimbraetal.,2004),(ZegelingandBlom,1992).
Anℎ-methodwasdevelopedforsolvingelectrochemistryproblems withlargespatialgradientsapplyingafinitedifferencemethod(Bieni- asz,2000).Laterthismethodwassuccessfullyusedforthesimulationof theNernst-Planck-PoissonequationsandNernst-Planckequationwith theassumption of electroneutrality((Bieniasz,2004a) and(Bieniasz, 2004b)).More examples abouttheℎ-refinement arefoundin (Lang, 1998,Wangetal.,2004,TrompertandVerwer,1991).
Probably themost significant adaptivemesh techniqueregarding ourworkis thealgorithmsuitableformodeling thetransientbehav- iorofthesamesystemwithcompletelydifferentboundaryconditions (Pˇribyletal.,2006).ThismethodisattachedtothecommercialFEM- LAB routines, which arebased on the finiteelement method. Their monitorfunctionisderivedfromthespatialderivativesofhydrogen,hy- droxylionconcentrationsandthepotential.Duetothetime-consuming evaluationofthemonitorfunctionandsuccessivemeshadaptation,the evaluationisnotcarriedoutateachtimeintegrationstep,theadapta- tionandconsecutiveevaluationtimeintervalisdeterminedbasedon theestimationoftransporttimes.
1.1. Aimofthiswork
Inthispaperwewouldlike toreportourempiricalmovingmesh method (𝑟-refinement), which was developed to model the time- dependentsalt-effectsofanelectrolytediode.Thealgorithmisattached tothecommercialCOMSOLequation-basedmodelingsoftware,which providesthespatialandtimediscretizationFEMalgorithmswithwell- definedinterfaces.Themainideaofthismethodis,thatinareaction- diffusionsystem,wherereactionfrontsmove, thelocalreactionrate indicatingthepresenceofanychemicalreactioncanbethebasisofan
Fig. 1.Schematicviewofastrongacid-basediode:Analkalinereservoir(filled bye.g.0.1MKOHsolution)andanacidicone(filledbye.g.0.1MHClsolution) areconnectedbyahydrogel.Δ𝜑>0meansreversebiaseddiode,theopposite isforwardbiased.
adaptivemeshrefinement.Theproblematicpartofsuchasystemisthe neighborhoodofthelocalchemicalreactions,wherethegradientsand thenumericalerrorofapproximationarethehighest.Thisideacould bethebasisof anyspatialdiscretization method, notonly thefinite elementmethod,theadvantageswouldbethesame:easyimplementa- tion,simplemonitorfunction,hencetherequiredmeshdensitywould bedeterminedinanempiricalwaynotdirectlyderivedfromtheerror ofapproximation.
2.Numericalsimulations 2.1. Themodel
Theso-calledacid-basediode(orelectrolytediode)isareaction- diffusion- ion-migrationsystem(NoszticziusandSchubert,1973,Schu- bertandNoszticzius,1977),where,duetotheapplied potentialand concentrationgradients,thechemicalspeciesmove,whileanacid-base reactionoccurs. Previouslysome highlynonlinearphenomenacalled positiveandnegativesalteffectsrelatedtothesalt-contaminationof theacid-basediodewerediscovered,thusitisstraightforwardtouse thenegativesalteffecttosensitivelydetectnonhydrogencations(Chun andChung,2015,Roszoletal.,2010,Kirschneretal.,1998).
Inanacid-basediodeaconnectingelement(eitherahydrogelora membrane)connectsanalkaline(KOH)andanacidic(HCl)reservoir, restrainingconvection,butallowingdiffusion,migrationandchemical reaction.(Theschematicviewofanacid-basediodeisshowninFig.1) Iftheacidic reservoirhasa negativepotentialin comparison tothe alkalineone(forwardbiaseddiode),fromthealkalinereservoircations (e.g.K+),fromtheacidiconeanions(e.g.Cl−)canmigrateintothegel cylinder,wheretheyformawell-conductingsalt(inthiscaseKCl).If theacidicreservoiristhepositiveside(reversebiaseddiode),hydrogen (H+)andhydroxil(OH−)ionscanmoveintothehydrogel,wherewater isformedfromtheirreaction,resultinginmuchsmallerconductivity.
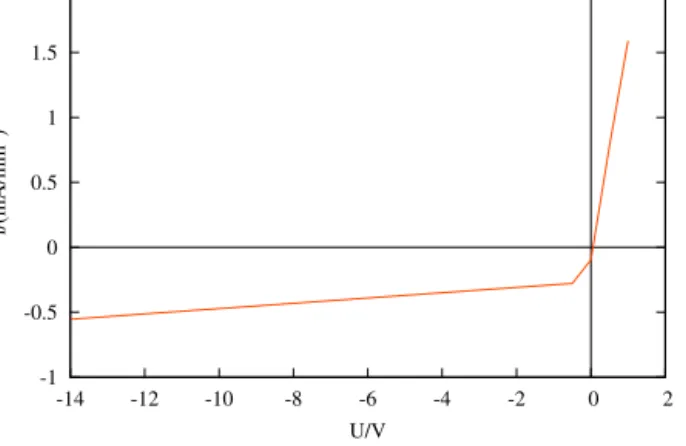
Mostgelscontain fixedweaklyacidicgroups.Thedissociationof themleadstofixednegativeions,inthepresentworkFA−isthefixed charge,andHFA denotesits protonatedform.The current - voltage characteristicofanelectrolytedioderesemblestothecharacteristicofa semiconductorone,meaningthediodecurrentdependsonthepolarity oftheappliedvoltage,asFig.2shows.
Ifthereservoirsofthediodearecontaminatedbyasalt(inthecase ofstrong diodeby KCl),theconcentrationof thecontaminating salt influencesthecurrentofthereversebiaseddiode:
• If only one reservoir is contaminated (usually the alkaline one duetohighersensitivity),thediodecurrentincreasesinanonlin- earway(Fig.3/a).Thisphenomenoniscalledpositivesalt-effect ((Kirschneretal.,1998,Heged˝usetal.,1999)).
• Ifonesideofthediodeisalreadycontaminated,andtheopposite reservoirofthediodegetscontaminatedaswell,surprisinglythe
Fig. 2.Simulatedvoltage–currentdensitycharacteristicofastrongacid-base diode.Concentrationsoftheelectrolytes:[KOH]=[HCl]= 0.1M.
Fig. 3.Calculatedcurrentdensityasthefunctionofsaltcontamination.(a):
positivesalteffect,(b):negativesalteffect.[KCl]ALand[KCl]ACdenotethesalt concentrationinthealkalineandacidicreservoirs,respectively.Theblackdot insubfigure(a)representsthealkalinesaltconcentrationusedforsubfigure(b).
[KCl]AL= 60mM,Δ𝜑= 10V.
diode currentdecreases byincreasedcounter saltcontamination (Fig.3/b).Thisphenomenoniscallednegativesalt-effect.
Bothpositiveandnegativesalteffectshadalreadybeeninvestigated for stationarystate (Kirschneret al., 1998, Roszoletal., 2010), we startedtofocusonthetime-dependentmodelingofsalt-contaminated diode (Konczetal.,2017),wherewemetunexpected computational complexity.
Atthisstageofinvestigation,themathematicalmodelofthediode isasystemofone-dimensionalpartialdifferentialequations(Roszolet al., 2010, Merkinet al., 2000, Heged˝uset al., 1996, Lindneret al., 2002).Thepreviouslydiscussedsimplificationsandassumptions((Iván etal.,2002))arekept(onedimensionalmodel,concentrationpolariza- tionandohmicpotentialdroponthereservoirareneglected).
Tocalculatetheconcentrationdistributionofthe𝑖-thcomponentthe mass-balanceequationisused,andtheNernst-Planckequationisused toobtainthecurrentdensity:
𝜕𝑐𝑖
𝜕𝑡 = −𝜕
𝜕𝑥 (
−𝐷𝑖𝜕𝑐𝑖
𝜕𝑥−𝑧𝑖𝐷𝑖𝐹 𝑅𝑇 𝑐𝑖𝜕𝜑
𝜕𝑥 )
+𝜎𝑖, (1)
where𝑐𝑖,𝑧𝑖and𝐷𝑖denotetheconcentration,chargenumberanddiffu- sioncoefficientofthe𝑖-thcomponent(H+,OH−,K+,Cl−,FA−,HFA), respectively.𝑇 isthetemperature,𝐹 istheFaradayconstant,𝑅isthe molargasconstant,𝜑denotesthespacedependentpotential, 𝑡isthe timecoordinateand𝑥isthespacecoordinatealongtheconnectingele- ment.𝜎𝑖denotesthereactionrateofthe𝑖-thchemicalcomponent:
𝜎H+=𝑘w(𝐾w−𝑐H+𝑐OH−) +𝑘f(𝐾f𝑐HFA−𝑐H+𝑐FA−), (2)
𝜎OH−=𝑘w(𝐾w−𝑐H+𝑐OH−), (3)
𝜎K+=𝜎Cl−= 0, (4)
𝜎FA−= −𝜎HFA=𝑘f(𝐾f𝑐HFA−𝑐H+𝑐FA−), (5) where𝐾w isthe equilibriumconstantof water dissociation,and𝑘w
denotestherateconstantofwaterrecombination.𝐾fisthedissociation constantand𝑘fdenotesthekineticconstantofthefixedcharge.
Poissonequation is used tocalculate thepotential profileof the diode:
𝜕
𝜕𝑥 (
−𝜀𝜕𝜑
𝜕𝑥 )
=𝐹( ∑
𝑖 𝑧𝑖𝑐𝑖)
, (6)
where𝜀denotesthepermittivityofwater.
InourmodelcalculationsDirichletboundaryconditionswereap- plied.Donnanequilibriumisusedtocalculatetheconstantboundary concentrationsinsidetheconnectingelementfromthefixedconcentra- tionsinthereservoirs.TheDonnanpotentialwasusedtocorrectthe electricpotential.
Unfortunatelywewerenotabletoreachconvergence,iftheabove discussedfullsystemofdifferentialequationsisapplied (massbalance equationsaresetupforeachspecies).Forstationaryinvestigationsthe concentrationof thefixedanions canbecalculatedfromtheequilib- rium,
𝑐FA−= 𝑐f
𝑐H+∕𝐾f+ 1, (7)
where𝑐fdenotestheestimatedtotalconcentrationofthefixedgroups (Ivánetal.,2004),thereforethemass-balanceequationisusedforthe mobileions(H+,OH−,K+,Cl−)only.
This assumption of quasi-stationarity was considered to be valid evenintimedependentcasesTheusageofthissimplification means thattheprotonationanddeprotonationofthefixedchargegroupsare consideredinstantaneous,thusthereactionratebelongingtothefixed groupsisapproximatedbyzero(𝜎FA−=𝜎HFA= 0).Theremainingreac- tionrateofH+is:𝜎H+=𝜎OH−=𝑘w(𝐾w−𝑐H+𝑐OH−).
Withthissimplificationitbecomespossibletosolvetheremaining PDEswithCOMSOL.
TheappliedvaluesoftheparameterscanbefoundinTable1.
2.2. SolutionmethodwithCOMSOL
ThesimulationswerecarriedoutbyusingthecommercialFEMsoft- warepackageCOMSOLMultiphysics4.3.Themodelwasdefinedfrom thefollowing interfaces: for the Nernst-Planck equation the “Trans- portofDilutedSpecies”and“ClassicalPDEs/Poissonequation”were applied.Afullmodelcalculationcanbesplitintotwomainsteps:af- tercomputingtheboundaryconditions,theinitialconditions(thisisa stationarystateofthediode)arecalculatedbyapreviouslydiscussed parametricstationarysolver(Roszoletal.,2010),whichisfollowedby thetime-dependentstudy.Thesolversettingsareadjustedunderthe
“Study”node,wherethetwostudystepsbelongingtothesolversare defined.Betweenthetwostudystepstheboundaryconditionsmustbe resetforthetransientvalue.(Inthecaseofstep1Computetoselected, inthecaseofstep2Computefromselectedcommandmustbeused.)
Forthenumerical integration of thetime-dependent ODEs(after spatialdiscretization) BDF method with SPOOLES linear solverwas appliedwiththefollowingsettings:automaticscaling,thetime-steps aredetermined freelyby thesolver. Toavoidnumerical oscillations crosswinddiffusionstabilizationtechniquewasapplied.Secondorder Lagrangeelementswereused.
ThekeyofFEMsimulationsistheappliedmesh.Thegradientsofthe concentrationsandpotentialarethelargestintheneighborhood ofthe so-calledreactionzone,thisisthehardesttocalculatepartofanacid- basediode.Toachieveconvergence,inthiszoneextremelyfinemesh isrequired.Duringthetime-dependentstudythelocationofthereac- tionzoneischanging,henceanadaptivemeshingtechniqueishighly recommendedtoreducecomputationalcosts.
In COMSOL Multiphysics adaptive mesh refinementextension is availableforthetime-dependentstudies.Unfortunatelythisbuilt-intool
Table 1.Modelparameters(Marcus(1997)).
𝑐KOH(moldm−3) Concentration of base 0.1
𝑐HCl(moldm−3) Concentration of acid 0.1
𝐷H+(m2s−1) Diffusion coefficient of protons 9.31 × 10−9 𝐷OH−(m2s−1) Diffusion coefficient of hydroxyl ions 5.28 × 10−9 𝐷K+(m2s−1) Diffusion coefficient of potassium ions 1.96 × 10−9 𝐷Cl−(m2s−1) Diffusion coefficient of chloride ions 2.04 × 10−9 𝐷FA−(m2s−1) Diffusion coefficient of the fixed charge 0 𝐷HFA(m2s−1) Diffusion coefficient of the protonated fixed group 0
𝜑L(V) Electric potential on the L boundary 0
𝜑R(V) Electric potential on the R boundary 10 − 20
𝑙(m) Length of the gel 1 × 10−3
𝑧H+ Charge number of protons 1
𝑧OH− Charge number of hydroxyl ions −1
𝑧K+ Charge number of potassium ions 1
𝑧Cl− Charge number of chloride ions −1
𝑧FA− Charge number of the fixed charge −1
𝑧HFA Charge number of the protonated fixed charge 0
𝐹(Cmol−1) Faraday constant 9.6487 × 104
𝑅(Jmol−1K−1) Molar gas constant 8.314
𝑇(K) Temperature 298.15
𝜀(AsV−1m−1) Permittivity of water 6.954 × 10−10 𝑘w(m3mol−1s−1) Kinetic constant of water recombination 1.3 × 108 𝐾w(mol2m−6) Ionic product of water 1 × 10−8 𝑐f(molm−3) Concentration of the fixed charge 4 𝑘f(m3mol−1s−1) Kinetic constant of the fixed charge 6 × 106 𝐾f(molm−3) Dissociation constant of the fixed charge 1 × 10−1
Fig. 4.The general operation of adaptive meshing algorithms.
andtheCOMSOL’sMovingMeshinterfaceprovedtobeinadequatefor ourpurposes.Thusweattachedanownadaptivealgorithmtooursim- ulationframework.(CarryingoutmodelcalculationsinCOMSOLGUI inseriesiscumbersome,thereforeaneasy-to-useframeworkwasdevel- opedinJavausingtheCOMSOLJavaAPI.)
ForthegeneraloperationofadaptivemeshingalgorithmsseeFig.4.
Thenumericalsolvermustbemonitored,andattheendofeveryiter- ationstepitmustbedecided,whethertheiterationcanbecontinued (meshisstillsatisfying)oranewmeshshouldbegenerated.Incaseofa newmeshthelastsolutionwiththeoldmeshisinterpolatedtothenew one,andthenumericalsolverisrestarted.InCOMSOLtheFEMalgo- rithmsareconsideredtobe“blackboxes”,thatisthenumericalmethod canbereachedonlyviatheinterfacesprovidedbyCOMSOL.Tostop andmonitorthenumericalsolver,astopconditionisformulatedapply- ingtheso-called“DomainPointProbe”.Withapointprobeexpression afunctionofvariablesofthedifferentialequationatapre-definedlo- cationisevaluated.
Atthetwoedgesofthereactionzone,insidethedensemeshzone, twopointprobeexpressionsaredefined.Thevaluesof themarethe absolutevalueoftheacid-basereaction’slocalreactionrateatthegiven location:
𝑝𝑝𝑣=||𝜎H+||=||𝜎OH−||=||𝑘w(𝐾w−𝑐H+𝑐OH−)||. (8) Thenthestopconditionisthefollowing:ifoneofthepointprobeex- pressionsdiffersignificantlyfromzero(𝑝𝑝𝑣>1.3⋅10−3mol
dm3s),thesolver
isstopped.This𝑝𝑝𝑣valuemeansthesolverstopswhen𝑐H+⋅𝑐OH−> 𝐾w, wegetthisthresholdbytrialanderror.Afterthat,fromthebehavior ofthereactionzoneanewmeshisgenerated.WithCOMSOLJavaAPI inthecaseofone dimensionalgeometryonlythevertexcoordinates arecalculated, theedges aretrivial(how thevertexesareconnected toanelement).InCOMSOLthesolutionofthetime-dependentsolver iscopied(Copysolution)toretaintheresults,andthedomainpoint probesarerelocatedbeforethetime-dependentsolverisrestarted.
2.3. Adaptivealgorithmwithgridfunction
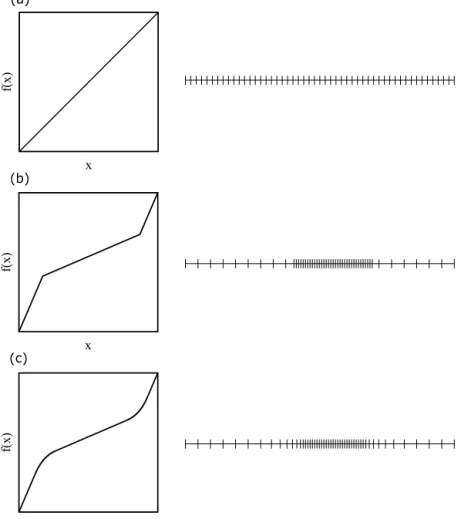
Theso-calledgridfunctionwasselectedtodefinethemesh.An𝑥→ 𝑓(𝑥)gridfunctiondefinestherealmesh’scoordinatesasafunctionof theequidistantmesh’scoordinates.Somegridfunctionswithschematic onedimensionalmeshesareshowninFig.5.
Thebasisofourselectedgridfunctionisanequidistantmeshwitha refinedzone(Fig.5/b),wherethevicinityofthereactionzoneiscov- eredbyanextremelyfinemesh.Toavoidnumericalproblemsarising fromsuddenchangesinthesizeofneighboringelements,inthevicin- ityofthestraightline’sintersectionsexponentialsmoothingfunctions areapplied.Formoredetailsseethe1stsectionoftheSupplementary Information.
Theadaptivealgorithmbasedonthegridfunctionhasthefollowing parameters:
• 𝑁:totalnumberofmeshpoints
• 𝑦A,𝑦B:inreal-meshcoordinatestheinitiallimitsoftheextremely finemeshzone
• 𝑅:ratioofthenumberofpointsbetween𝑦Aand𝑦Bandthetotal numberofgridpoints
• 𝛿: widthof theexponentialsmoothing partin equidistant mesh coordinates
• 𝑑: distancebetween 𝑦A or 𝑦B andits correspondingpointprobe expression
• Δ𝑦:howmuchthecontrolpoints(𝑦A,𝑦B)ofthegridfunctionare shiftedintothedirectionofthereaction’smovement,afterthestop conditionisfulfilled,andthenextmeshisgenerated
Themainadvantageofapplyingagridfunctionis itseasyimple- mentation, simple mesh generation, and low time-demand of mesh
Fig. 5.Somegridfunctionswiththecorrespondingschematicmeshillustrations.(a)Equidistantmesh.(b)Equidistantmeshwitharefinedzone.(c)Theapplied gridfunctionwithexponentialsmoothing.
construction. Forthe source codesee https://github.com/vikikoncz/ adaptive_FEM_based_reaction_zone.
3. Resultsanddiscussion
Wewouldliketoshowtheabilityoftheadaptivemeshalgorithm tosimulatethetime-dependentbehaviorofanacid-basediode.Inthe presentworkwedemonstrate theoperationofthealgorithmincase of time-dependentpositive salteffect withthefollowingsettings. As initialstatethesalt-freestationarydiodewasused.Thecontaminating salt was addedtothealkaline reservoirat 𝑡= 0,[KCl]AL,t= 60mM.
Approximatelyafter100sthenewsteadystatewasattained,thus150 swassimulatedafterthesystemwasperturbedbyKClcontamination.
Ourworkispresentedinthreesubsections.First,inSection3.1the appearanceofthereactionzoneisinvestigated.Wewouldliketoshow, that thelocation of thechemical reaction (where thereaction ratio differssignificantlyfromzero)isveryclosetothelocationwherethe gradientsoftheconcentrationandpotentialarethehighest.
Then,weinvestigatedtheabsoluteandrelativeerrorofapproxima- tiononthemodeleddomainforvalidationpurposes(Section3.2).
Finally,we discussthe effectof different meshes onthe error of approximationandsimulationtime(Section3.3).
3.1. Reactionzone
Thelocalreactionrateoftheacid-basereactionis𝜎H2O= −𝑘w(𝐾w− 𝑐H+𝑐OH−).Itisthelinearfunctionof[H+]⋅[OH−],whichinequilibrium is atemperaturedependentconstant.Fig.6shows the localreaction rate(𝜎H2O)alongthegelcylinderinastationarysalt-freediode.𝜎H2O
Fig. 6.Thelocalreactionrate(𝜎H2O= −𝑘w(𝐾w−𝑐H+𝑐OH−))ofwaterrecombina- tionintheuncontaminateddiode.
isdifferent fromzeroonlyat thereaction zone,wheretheacid-base reactionoccurs.
Fig.7showssomecalculated𝜎H2Oprofilesofatime-dependentpos- itivesalt effect. Atacertaintime𝜎H2Ois differentfrom zeroinone regiononly,certainlythisisthereactionzone.
Asthealkalinereservoir(lefthandside)isapproachedbythereac- tionzone,theacid-basereactionbecomesfasterandfaster(thepeakof 𝜎H2Oincreases).Thereactionzonekeepsitspositionafterabout10s, however,thereactionratecontinuesincreasing.
Thelookofthereactionzoneatfourselectedmomentsoftimeis showninFig.8.Whiletheacid-basereactiongetsfaster,theextension ofthereactionzoneshrinks.Inthenewsteadystateitswidthisonly
Fig. 7.Thelocalreactionrate(𝜎H2O= −𝑘w(𝐾w−𝑐H+𝑐OH−))ofwaterrecombina- tionatdifferentmomentsoftime.
thethirdoftheuncontaminatedone.Theextensionofthereactionzone iscalculatedfromthehalf-widthofthe𝜎H2Opeak.
Intheuncontaminatedsteadystatethepositionsoftheextremaof spatialderivativesarevery closetothelocationwheretheacid-base reactionoccurs.Thesederivativesmovetogetherwiththechemicalre- action.Thelocalextremaofconcentrationderivativesareincreasing(in absolutevalue),whilethederivativeofpotentialchangesonlyslightly.
Thesituationisthesameinthecaseofsecondspatialderivatives.
3.2. Validation
Inthecaseofasystemofnonlinearpartialdifferentialequations, itispossible,thatthenumericalsolverfindsaphysicallyinvalidso- lutioneventhoughitsatisfiesthetolerancecriteria(LeVeque(1992)).
Weusedaposteriorierrorindicatorstocharacterizetheprecisityofthe numericalsolutions.
Ameaningfulmeasureofthecomputationalerrorcanbegivenon (𝑥𝑖,𝑥𝑖+1)asfollows:
𝑥𝑖+1 𝑥∫𝑖
(̃𝑦(𝑥) −𝑦(𝑥))2 𝑑𝑥=𝑅(𝑥,𝐚), (9)
where𝑦(𝑥)denotestherealsolution ofthedifferentialequationand
̃𝑦(𝑥)=∑𝑛
𝑗=1𝑎𝑗⋅Φ𝑗(𝑥)isthefiniteelementapproximation.Inpractice, therealsolutionisunknown,therefore,anothererrorindicatorhasto befound.
Ifthecoefficients𝑎𝑗ofthebasisfunctionsareavailable,substituting themintotheoriginalPDE’sleadstoanerrorindicatoroneachelement.
UnfortunatelyduetoCOMSOL’s‘blackbox’approachitisneither possibletogettheLagrangian coefficients,northeerrorcalculatedby COMSOL.
Tofindametricforcomparingdifferentsolutions,anownmethod hadtobedeveloped.ItispossibletoexportfromCOMSOLtheinterpo- latedsolution,andthespatialandtimederivativesofthevariablesof theequationatthepredefinedpointsinagivenmoment.(FromCOM- SOLJavaAPItheso-called‘Interp’functionwasappliedtoretrievethe numericalresults.)Ourmethodisthefollowing:duringtimedevelop- mentateverydiscretemomentoftimetheapproximatedvalueofthe variables,the1stand2ndspatialderivativesandthetime-derivatives areexportedatthemeshpoints.Thesevaluescanbesubstitutedtothe fivedifferentialequations(fourmassbalancesandthePoisson)atevery momentandlocation,leadingtofive(absolute)residualvalues𝑒𝑟𝑟abs,𝑖. Asanexample,thecalculatedabsoluteresidualvaluesoftheH+’s massbalanceequation,andthePoissonequationareshowninFigs.9 and10.
Theabsolute error in itselfdoesnot tellusthe wholepicture,it hastobecomparedtotheabsolutevaluesofthevarioustermsinthe givenequation.Forthisreasonweusetherelativeerrordefinedinthe followingway:
𝑒𝑟𝑟rel,𝑖= 𝑒𝑟𝑟abs,𝑖
|||𝜕𝑐𝜕𝑡𝑖|||+||
||−𝜕
𝜕𝑥
(
−𝐷𝑖𝜕𝑐𝜕𝑥𝑖)|
|||+||
||−𝜕
𝜕𝑥
(
−𝑧𝑖𝐷𝑖𝐹
𝑅𝑇 𝑐𝑖𝜕𝜑𝜕𝑥)|
|||+||𝜎𝑖|| (10) 𝑒𝑟𝑟rel,Poisson= 𝑒𝑟𝑟abs,Poisson
||||𝜕𝑥𝜕 (
−𝜀𝜕𝜑𝜕𝑥)|
|||+||
||𝐹( ∑
𝑖𝑧𝑖𝑐𝑖)|
|||
(11)
Despiteofthelarge absoluteerrorintheneighborhoodofthere- actionzone,therelativeerrors (Figs.11and12) seemtobe mostly acceptable.However,averysharpregionexistsinthemiddleof the reactionzone,wheretheapproximationerrorofthePoissonequation becomes0.65.
3.3. Theeffectofthegridfunction’sparametersontheerrorof approximationandonthesimulationtime
Tocomparethedifferenttypesofmeshes,thesimulationtimemea- suredinanisolatedvirtualmachine(fordetailsseethe2ndsectionof SupplementaryInformation)wasmonitored,andtheerrorofapproxi- mationwasestimated.Unfortunately,thenumberofiterationsarenot measurableviaCOMSOL,thuswehadtomeasuretherunningtimeof thesimulations.It isalso importantinpractice, butdepends on the hardware.ThebasicstepsofanadaptiveFEMsimulationwiththemea- suredrunningtimevaluesarefoundinthe4thsectionofSupplementary Information.About97%ofthetotalsimulationtimegoestothetime- steppingandmeshinterpolation, thus therunningtimeoftheother stagesofFEMsimulationisnegligible.
Insteadofplottingtheresidualsateverymomentoftime(likein Section3.2),itisbettertouseacumulatederrorindicatortodescribe theapplicabilityofthemesh.Toconstructsuchanaposteriorierror indicator,weconsiderequation (1)asasystemofconservation laws coupledwiththereactivesourceterms(𝜎H+,𝜎OH−,0,0).Usingthecor- respondingtheoryforthenon-linearconservationlawsinTheorem5.2 in(Cockburn,1999),astraightforwardchoicefortheerrorestimation ofthe𝑗-thcomponentinequation(1)canbecalculatedasfollows:
𝑒𝑟𝑟𝑗=
𝑇
∫
0 𝐿
∫
0
|𝑒𝑟𝑟abs,𝑗|𝑑𝑡𝑑𝑥+
√√
√√
√√max
𝑡∈[0,𝑇]𝑆(𝑐𝑗)(𝑡)⋅
𝑇
∫
0
𝑆(𝑐𝑗)(𝑡)𝑑𝑡⋅√
𝐷𝑗, (12)
where 𝑒𝑟𝑟abs,𝑗 denotes the𝑗-th componentscalculated residual, and 𝑆(𝑐𝑗)(𝑡)isthesumoftheconcentrationabsolutedifferences between thegridpointsontheentiremodeleddomain.Wefound,thattheterm containing𝑆isalmostnegligiblecomparedtothetimeandspatialin- tegraloftheresiduals.Furthermore,itwasfoundthatthistermvaries within1%inthecaseofeverymeshsetting(ifconvergenceisachieved), thusitcanbeneglectedwhencomparingmeshes.
Forasinglecumulativeerrorindicatortheerrorestimationofthe fourcomponentsissummed andnormedby𝑇= 150s (duetothetime integration):
𝑒𝑟𝑟𝑜𝑟=𝑒𝑟𝑟H++𝑒𝑟𝑟OH−+𝑒𝑟𝑟K++𝑒𝑟𝑟Cl−
𝑇 . (13)
Inthefollowingtablesandfiguresonlythiscumulativevalueisused tocomparedifferentmeshes.
First,theefficiencyof theadaptivemeshing algorithmisshowed withcomparisontotwofixedmeshsimulations.Afterthat,theeffects ofthegridfunctionparametersareexplored,andwetriedtofindan optimalparametercombinationconsideringthesimulationtimeandthe errorofapproximation.(Inthisstudytheparametersarefixedduringa simulation.)Finally,weinvestigatedhowthecontinuousmodification ofthegridfunction’sparametersinfluencesthesimulation.
3.3.1. Fixedmeshvsadaptivemesh
Theperformanceofanequidistantmesh(namedfixed(1)),afixed meshwithadensezone(fixed(2))andthreemovingmesheswiththe proposedgridfunctionwerecompared(Table2).Therunningtimeof
Fig. 8.Reactionfronts:spatialgradientsandthelocalreactionrateofacid-basereactionindifferentmomentsoftime.Inthefirstcolumnthelocalreactionrateand the1stspatialderivativesareshownatt=0s(a),t=3s(c),t=5s(e)andt=20s(g).Inthesecondcolumnthelocalreactionrateandthe2ndspatialderivatives areshownatt=0s(b),t=3s(d),t=5s(f)andt=20s(h),respectively.Pleasenote:thederivativesandthelocalreactionratearescaled,thusnounitisshown inthisfigure,becauseweareonlyinterestedinthepositionsofpeaks,nottheirmagnitude.
Fig. 9.TheabsoluteerroroftheH+ionsmassbalanceequation(𝑒𝑟𝑟abs,H+)at 𝑡= 3s.Theparametersoftheappliedmesharethefollowing:𝑁= 10000,𝑦A= 0.192,𝑦B= 0.202,𝑑= 0.002,Δ𝑦= 0.002,𝛿= 0.01,𝑅= 0.8.
Fig. 10.Theabsoluteerrorof thePoissonequation(𝑒𝑟𝑟abs,Poisson)at𝑡= 3s.
Theparametersoftheappliedmesharethefollowing:𝑁= 10000,𝑦A= 0.192, 𝑦B= 0.202,𝑑= 0.002,Δ𝑦= 0.002,𝛿= 0.01,𝑅= 0.8.
Fig. 11.TherelativeerroroftheH+ionsmassbalanceequation(𝑒𝑟𝑟rel,H+)at𝑡= 3s inthereactionzone.Theparametersoftheappliedmesharethefollowing:
𝑁= 10000,𝑦A= 0.192,𝑦B= 0.202,𝑑= 0.002,Δ𝑦= 0.002,𝛿= 0.01,𝑅= 0.8.
Fig. 12.TherelativeerrorofthePoissonequation(𝑒𝑟𝑟rel,Poisson)at𝑡= 3s inthe reactionzone.Theparametersoftheappliedmesharethefollowing:𝑁= 10000, 𝑦A= 0.192,𝑦B= 0.202,𝑑= 0.002,Δ𝑦= 0.002,𝛿= 0.01,𝑅= 0.8.
Table 2. The measured running time and calculated errorofapproximation(error)in the caseoffixedandadaptivemeshes. The fixed(1)meshisanequidistantmesh,fixed(2) isa fixedmesh witha densezone. Param- etersofthegridfunctionarethefollowing:
𝑦A= 0.192,𝑦B= 0.202,𝑑= 0.002,Δ𝑦= 0.002, 𝛿= 0.01,𝑅= 0.8.
Mesh type N Time (min) error
fixed(1) 100000 3648 147
fixed(2) 21000 532 73
adaptive 500 15 183
adaptive 1000 35 97
adaptive 2000 42 49
adaptive 10000 222 10
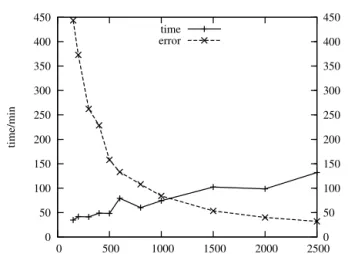
theequidistantmesh(10000gridpoints)is thebiggest,andapprox- imatelythesameprecision can beachieved usinganadaptive mesh with500- 1000gridpointsdependingonthegridfunctionparameters.
Thefixed(2)meshhasthesameresolutionastheequidistantmesh (fixed(1))initsdensepart,whichcoversthatpartofthegel,wherethe reactionzonecanoccur.Theremainingpartofthegeliscoveredbyan 80timeslessdenseequidistantmesh,andthetwozonesareconnected byanexponentialsmoothingpart.
Despitethesameresolutioninthereactionzone,theerrorofapprox- imationisalmosthalvedinthecaseoffixed(2),duetotheconvergence
Fig. 13.Themeasuredrunningtime andtheerrorofapproximationasthe functionofthenumberofgridpoints.Parametersofthegridfunctionarethe following:𝑦A= 0.192,𝑦B= 0.202,𝑑= 0.002,Δ𝑦= 0.002,𝛿= 0.01,𝑅= 0.8.
criteriaofCOMSOL’snonlinearsolver.Intheaggregatederrorevalu- atedbyCOMSOLaftereveryiterationstep,theerroronanelementis notweightedbytheelement’ssize.ThusinCOMSOL’saggregatederror, inthecaseofequidistantmeshtheelementsbelongingtothereaction zoneareunderweightedcomparedtothefixed(2)mesh.(Furtherdetails aboutCOMSOL’sconvergencecriteriacanbefoundinthe3rdsection oftheSupplementaryInformation.)
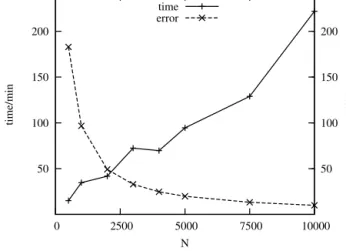
Themostaccuratesolution isachievable byamovingmeshwith asmany gridpointsasthedesiredrunningtimeallows.(Inourcase maximum10000meshelementswereused.)Inthecaseofonespecific adaptivemeshparametersettingthemeasuredrunningtimeandthe errorofapproximationasthefunctionofthenumberofgridpointsare showninFig.13.(Meshtrajectoriesofthismovingmeshcanbefound inthe7thsectionoftheSupplementaryInformation.)
Ifthenumberofgridpointsincreases,theapproximationerrorde- creasesinahiperbolicway.Therunningtimecanshowdiscrepancies, itdoesnotincreasemonotonicallyasafunctionof𝑁.
Theconvergenceratesofthesediscretizationschemes (fixed(2),and adaptive)arecomparedinthe6thsectionoftheSupplementaryInfor- mation.
3.3.2. Theeffectofthegridfunctionparameters
Inthissectionwepresentourfindingsaboutsystematicinvestiga- tionsoftheeffectofgridfunctionparametersontheperformanceof themovingmeshalgorithm.Pleasenote,duringonesimulationthepa- rametersofthegridfunctionwerekeptconstant.Duringthediscovery oftheparameters𝑁= 2000waschosen,becausewefounditagood compromisebetweenrunningtimeanderrorofapproximation(seeTa- ble2).
First,theeffectofΔ𝑦wasinvestigated(Fig.14).Itdoesnotinflu- encetheerrorofapproximationsignificantly,however,themeasured simulationtimedepends onit.ThesmallertheΔ𝑦is,thehigher the runningtimebecomes.Thefrequentmeshadaptationneedstoomuch interpolationbetweenthedifferentmeshes.
Withinawiderangethegridfunctionparametercalled𝑅hasvery similareffect ontheerror andsimulation time(Fig. 15).If𝑅 istoo small,thesolverjust cannotconverge(becausetheresolutionis not highenoughinthereactionzone).Certainly,theprecisevaluedepends on thenumberof grid points,and theotherparameters of thegrid function,e.g.if𝑁= 2000, 𝑅mustbe atleast 0.05.However,in this casetheapproximationerrorishuge(2600).(Forthisreasonitisnot showninFig.15.)Theerrorofapproximationhasanoptimumaround 𝑅= 0.94,if𝑅islarger,toofewgridpointsappearoutsidethereaction zone.
Fig. 14.Themeasuredrunningtimeandtheerrorofapproximationasthe functionoftheΔ𝑦parameter.Parametersofthegridfunctionarethefollowing:
𝑦A= 0.191,𝑦B= 0.203,𝑑= 0.003,𝑅= 0.8,𝛿= 0.01,𝑁= 2000.
Fig. 15.Themeasuredrunningtimeandtheerrorofapproximationasthe functionofthe𝑅parameter.Parametersofthegridfunctionarethefollowing:
𝑦A= 0.191,𝑦B= 0.203,𝑑= 0.003,Δ𝑦= 0.002,𝛿= 0.01,𝑁= 2000.
Dependingonthewidthoftheextremelyfinemeshzone,andon thegridfunctionparameters,anoptimal𝛿exists,wheretheerrorof approximationisthesmallest(Fig.16).If𝛿increasesfromzero,firstthe errorofapproximationdecreases,duetosmoothertransitionofelement size.When𝛿becomescomparabletothesizeofthedensemeshzone, thenumberofmeshelementsinthereactionzonestartstodecrease, resultinginincreasederrorofapproximation.
Duetothetimedemandoftoofrequentmeshadaption,anddueto convenience,densemeshwasusuallyusedonalargeenvironmentof thereactionzone.(Itmeans,𝑦B−𝑦Aisatleastfivetimesbigger,than thereactionzone’sextension.)Theimpactofthedistancebetween𝑦A
and𝑦B wasinvestigated(Fig. 17) toseewhethercoveringasmaller environmentwithdensemeshstillgivessatisfactoryresults.Theerror of approximation increaseswith thisdistance (because lessgrid ele- mentscoverthereactionzone).Usuallysmallererrorcomeswithlower runningtime,however,heretheoppositehappens:increasederrorde- creasestherunningtime.
Theminimalreasonabledistancebetween𝑦Band𝑦Aistheextension ofthereactionzone:underthislimitconvergenceproblemsappear.In thecaseofminimal𝑦B−𝑦Atheerrorandtherunningtimeasthefunc- tionof𝑁isshowninFig.18.Toreachconvergencewiththesesettings, theminimumnumberofrequiredelementsis𝑁= 150,althoughinthis casetheerrorofapproximationisaround450,andtheprofilesarenot
Fig. 16.Themeasuredsimulationtimeandtheerrorofapproximationasthe functionofthe𝛿parameter.Parametersofthegridfunctionarethefollowing:
𝑦A= 0.191,𝑦B= 0.203,𝑑= 0.003,Δ𝑦= 0.002,𝑅= 0.8,𝛿= 0.01,𝑁= 2000.
Fig. 17.Themeasuredrunningtime andtheerrorofapproximationasthe functionofthedistancebetween𝑦Aand𝑦B.Parametersofthegridfunctionare thefollowing:𝑑= 0.0005,Δ𝑦= 0.0005,𝑅= 0.8,𝛿= 0.01,𝑁= 2000.
Fig. 18.Themeasuredrunningtime andtheerrorofapproximationasthe functionofthe𝑁.Parametersofthegridfunctionarethefollowing:𝑦A= 0.196, 𝑦B= 0.199,𝑑= 0.0005,Δ𝑦= 0.0005,𝑅= 0.8,𝛿= 0.01.Thedensemeshbarely coversthereactionzone.
smoothenough.If𝑁= 500,thealgorithmhasverysimilarperformance, as𝑁= 2000withthewidelycoveredzone.
Table 3. The measured running timeandtheerrorofapproxima- tionin thecaseof movingmesh algorithmswithdifferentcontinu- ouslymodifiedgridfunctions.Ini- tialvaluesofthegridfunctionpa- rameters: 𝑁= 10000, 𝑦A= 0.191, 𝑦B= 0.203,𝑑= 0.003,𝑅= 0.8,Δ𝑦= 0.002, 𝛿= 0.01. In the method calledCMG1onlythedistancebe- tween𝑦B−𝑦A,𝑑,andΔ𝑦arede- creased.InthemethodcalledCMG 2𝑅isalsodecreased,furthermore inthemethodcalledCMG3even 𝛿isdecreased.InthecaseofNMG (non-modifiedgridalgorithm)the initialvaluesoftheparametersare the same, and arekept constant duringthesimulation.
Mesh type Time (min) error
CMG 1 233 7
CMG 2 235 14
CMG 3 257 17
NMG 283 11
3.3.3. Continuousmodificationoftheparameters
Duringthetransitionofthediodetheextensionofthereactionzone shrinks.Inthenewsteadystate,it’sextensionisonethird,thaninthe saltfreediode(section3.1).Isitpossibletoreachbetterperformance byshrinkingthefinemeshzonewiththereactionzone?
Toalterthegridfunctioncontinuously,thepreviouslydiscussedal- gorithmismodified:ifthestopconditiondefinedinCOMSOLisfulfilled andtheiterationstops,thenthedependent variables(and itsspatial derivatives)mustberetrievedatthegridpoints.Thehalfwidthofthe localreaction ratepeakwas usedtoestimate thereactionzone’sex- tension,fromwhichnotonlythelocationofthefinemeshzone,but otherparametersofthegridfunctioncouldbere-calculated(forfurther detailssee5thsectionofSupplementaryInformation).Unfortunatelyif thetotalnumberofgridpointsistoosmall,theestimationofshrink- ageofthereactionzonebecomesunprecise,leadingtonon-converging mesh.Itmeans,thatinthecaseofsmall𝑁continuousmodificationof theparameterscanbenumericallyunstable.
The performance of algorithms with continuously modified grid (CMG)andanonmodifiedgrid(NMG)issummarized inTable3.The methodcalledCMG1,whereonly𝑦B−𝑦A,𝑑andΔ𝑦areshrinked,gives thesmallesterrorandsimulationtime.
4. Conclusionsandoutlook 4.1. Conclusions
We suggestan empiricalmoving meshalgorithm, which was at- tachedtothecommercialFEMsolverCOMSOL.Realizingthathigher reaction rateand higher concentrationusually appearclose to each other,thelocalreactionratewasusedtocontrolthemeshadaption.The proposed algorithmwas testedfor areaction-diffusion systemcalled acid-basediode,andtheparameterswerefine-tunedforthiscase.Dur- ingthetime-dependentmodelingoftheso-calledsalteffectsinthissys- temweexperiencedhighcomputationalcomplexity,thusanadaptive meshingalgorithmwasnecessarytoreducethetimeofthesimulation.
Wefound,thatusingthelocalreactionrateinsteadofthespatialderiva- tivesmakestheimplementationofthealgorithmveryeasyandsimple.
4.2. Outlook
Themovingmeshalgorithmcanbeextendedtohandletheappear- ance anddisappearance of thereactionzones.Ifthetime-dependent solverisstopped,oraftereveryiterationstep,themodeled domaincan
bescannedwithamonitorfunction,whichevaluatesthelocalreaction rates,andmarksthelocationofthereactionzone.Fromthelocationsa newgridfunctioncanbecreated,inwhichthelesssteepstraightseg- mentsofthepiecewisegridfunctioncorrespondtothereactionzones.
(Theexponentialsmoothing,andhandlingoftooclosereactionzones needtobedealtwith.)
Unfortunately,astraightforwardextensionoftheproposedprocess fortwo-dimensionalgeometricsetupseemstobeimpossible.Forsuch cases, thegrid-transformationbetween consecutivetime stepsis ob- tainedfrom thesolution ofso-called movingmeshPDE (Zegelinget al.,2005).Themeshhastoberegularizedinmanycasesandaninter- polationisalsonecessarybetweentwoconsecutivemeshes.Formany applications,however,one-dimensionalsituationsarealsoofgreatim- portance.
Furthermore,thelocalreactionratecanbeusedasa‘monitorfunc- tion’ not only in 𝑟-refinement, but in ℎ-refinement, 𝑝-refinement or ℎ𝑝-refinementmethodsaswell.Insuchcases,intheneighborhood of thechemicalreactionmeshelementscanbeadded(andlaterremoved), orthepolynomialdegreeof thebasisfunctioncanbe increased(and laterdecreased).Thesetechniquesareeasiertoextendtotwoormore dimensionalproblemssimulatedbyfiniteelementorfinitedifference methods.
Declarations
Authorcontributionstatement
ViktóriaKoncz&KristófKály-Kullai:Conceivedanddesignedthe analysis;Analyzedandinterpretedthedata;Wrotethepaper.Ferenc Izsák:Conceivedanddesignedtheanalysis;Wrote thepaper. Zoltán Noszticzius:Contributedanalysistoolsordata;Wrotethepaper.
Fundingstatement
ThisworkwassupportedbyOTKAGrantK131425andbytheELTE InstitutionalExcellenceProgram(1783-3/2018/FEKUTSRAT)through theHungarianMinistryofHumanCapacities.
Dataavailabilitystatement
Datawillbemadeavailableonrequest.
Declarationofinterestsstatement
Theauthorsdeclarenoconflictofinterest.
Additionalinformation
Supplementarycontentrelatedtothisarticlehasbeenpublishedon- lineathttps://doi.org/10.1016/j.heliyon.2020.e05842.
Acknowledgements
TheauthorsthankA.Patariczaforhelpfuldiscussions.
References
Bieniasz,L.K.,2000.Useofdynamicallyadaptivegridtechniquesforthesolutionofelec- trochemicalkineticequations:part5.Afinite-difference,adaptivespace/timegrid strategybasedonapatch-typelocaluniformspatialgridrefinement,forkineticmod- elsinone-dimensionalspacegeometry.J.Electroanal.Chem. 481(2),115–133.
Bieniasz,L.K.,2004a.Useofdynamicallyadaptivegridtechniquesforthesolutionof electrochemicalkineticequations.Part14:extensionofthepatch-adaptivestrategy totime-dependentmodelsinvolvingmigration-diffusiontransportinone-dimensional spacegeometry,andit.J.Electroanal.Chem. 565(2),251–271.
Bieniasz,L.K.,2004b.Useofdynamicallyadaptivegridtechniquesforthesolutionof electrochemicalkineticequations. Part15:patch-adaptivesimulationofexample transientexperimentsdescribedbyNernst–Planck–electroneutralityequationsinone- dimensionalspacegeometry.J.Electroanal.Chem. 565(2),273–285.
Cao,W.,Huang,W.,Russell,R.D.,2003.Approachesforgeneratingmovingadaptive meshes:locationversusvelocity.Appl.Numer.Math. 47(2),121–138.
Chun,H.,Chung,T.D.,2015.Iontronics.Annu.Rev.Anal.Chem. 8,441–462.
Cockburn,B.,1999.TheGraduateStudent’sGuidetoNumericalAnalysis’98.Springer, Berlin.
Coimbra,M.D.C.,Sereno,C.,Rodrigues,A.,2004.Movingfiniteelementmethod:appli- cationstoscienceandengineeringproblems.Comput.Chem.Eng. 28(5),597–603.
Farmaga,I.,Shmigelskyi,P.,Spiewak,P.,Ciupinski,L.,2011.Evaluationofcomputational complexityoffiniteelementanalysis.In:201111thInternationalConferencetheEx- perienceofDesigningandApplicationofCADSystemsinMicroelectronics(CADSM), pp. 213–214.
Heged˝us,L., Kirschner,N., Wittmann,M.,Simon, P.,Noszticzius,Z., Amemiya,T., Ohmori,T.,Yamaguchi,T.,1999.Nonlineareffectsofelectrolytediodesandtran- sistorsinapolymergelmedium.Chaos 9(2),283–297.http://www.ncbi.nlm.nih. gov/pubmed/12779826.
Heged˝us,L.,Wittmann,M.,Kirschner,N.,Noszticzius,Z.,1996.Reaction,diffusion,elec- tricconductionanddeterminationoffixedionsinahydrogel.Prog.Colloid&Polym.
Sci. 102,101–109.
Huang,W.,Ren,Y.,Russell,R.D.,1994.Movingmeshpartialdifferentialequations(MM- PDES)basedontheequidistributionprinciple.J.Numer.Anal. 31(3),709–730.
Iván,K.,Kirschner,N.,Wittmann,M.,Simon,P.L.,Jakab,V.,Noszticzius,Z.,Merkin, J.H.,Scott,S.K.,2002.Directevidenceforfixedionicgroupsinthehydrogelofan electrolytediode.Phys.Chem.Chem.Phys. 4(8),1339–1347.
Iván,K.,Wittmann,M.,Simon,P.L.,Noszticzius,Z.,Vollmer,J.,2004.Electrolytediodes andhydrogels:determinationofconcentrationandpKvalueoffixedacidicgroupsin aweaklychargedhydrogel.Phys.Rev.E 70(61),061–402.
Kirschner,N.,Heged˝us,L.,Wittmann,M.,Noszticzius,Z.,1998.Nonlinear“salt-effect”in anelectrolytediode.Theoryandexperiments.ACH- ModelsChem. 135,279–286.
Koncz,V.,Noszticzius,Z.,Roszol,L.,Kály-Kullai,K.,2017.Time-dependentmodelingof salt-contaminatedacid-basediodes.J.Electrochem.Soc. 164(4),H257–H265.http://
jes.ecsdl.org/lookup/doi/10.1149/2.1701704jes.
Lang,J.,1998.AdaptiveFEMforreaction—diffusionequations.Appl.Numer.Math. 26 (1–2),105–116.
LeVeque,R.J.,1992.NumericalMethodsforConservationLaws.Birkhäuser,Basel.
Lindner,J.,Šnita,D.,Marek,M.,2002.Modellingofionicsystemswithanarrowacid–
baseboundary.Phys.Chem.Chem.Phys. 4(8),1348–1354.
Marcus,Y.,1997.IonProperties.MarcelDekker,NewYork.
Merkin,J.H.,Simon,P.L.,Noszticzius,Z.,2000.Analysisoftheelectrolytediode.Electro- diffusionandchemicalreactionwithinahydrogelreactor.J.Math.Chem. 28,43–58.
Noszticzius,Z.,Schubert,A.,1973.“Electrolytediode”analysisofisothermaltransport processesintheinterfacesofaqueoussolutions.Period.Polytech.,Chem.Eng. 17, 165–177.
Pˇribyl,M.,Šnita,D.,Kubíˇcek,M.,2006.Adaptivemeshsimulationsofionicsystemsin microcapillariesbasedontheestimationoftransporttimes.Comput.Chem.Eng. 30 (4),674–685.
Roszol,L.,Várnai,A.,Lorántfy,B.,Noszticzius,Z.,Wittmann,M.,2010.Negativesalt effectinanacid-basediode:simulationsandexperiments.J.Chem.Phys. 132(6), 064902.
Schubert,A.,Noszticzius,Z.,1977.Electrolytediode- anexperimentalstudypolarization phenomenaatthejunctionoftheaqueoussolutionsofanacidandabase.PartII.
Period.Polytech.,Mech.Eng. 21,279–283.
Schwab,C.,1999.p- andhp- FiniteElementMethods.ClarendonPress,Gloucestershire.
Trompert,R.A.,Verwer, J.G.,1991.A static-regriddingmethodfortwo-dimensional parabolicpartialdifferentialequations.Appl.Numer.Math. 8(1),65–90.
Wang,R.,Keast,P.,Muir,P.,2004.Ahigh-orderglobalspatiallyadaptivecollocation methodfor1-DparabolicPDEs.Appl.Numer.Math. 50(2),239–260.
Zegeling,P.A.,Blom,J.G.,1992.Anevaluationofthegradient-weightedmoving-finite- elementmethodinonespacedimension.J.Comput.Phys. 103(2),422–441.
Zegeling,P.A.,deBoer,W.D.,Tang,H.Z.,2005.Robustandefficientadaptivemoving meshsolutionofthe2-DEulerequations.Contemp.Math. 383,375–386.
Zegeling,P.A.,Keppens,R.,2001.AdaptiveMethodofLinesforMagnetohydrodynamic PDEModels.AdaptiveMethodofLines,pp. 117–137.
Zegeling,P.A., Kok,H.P.,2004. Adaptive movingmesh computationsfor reaction- diffusionsystems.J.Comput.Appl.Math. 168(1–2),519–528.
Zimmerman,W.B.J.,2006.MultiphysicsModelingwithFiniteElementMethods.World ScientificPublishingCompany,Singapore.