4th International Workshop on the Biology of Fish Gametes 2013
128
S2-O-07
SYSTEMATIC OPTIMIZATION OF CONDITIONS FOR THE CRYOPRESERVATION OF EURASIAN PERCH (Perca fluviatilis) SPERM
Gergely Bernáth1*, Daniel arski2, Sławomir Krejszeff2, Katarzyna Pali ska- arska2, Tímea Kollár1, Zoltán Bokor1, Dariusz Kucharczyk2, Béla Urbányi1, Ákos Horváth1
1 Department of Aquaculture, Szent István University, H-2100, Gödöll", Hungary
2 Department of Lake and River Fisheries, University of Warmia and Mazury, Olsztyn, Poland
* Corresponding author: Tel.: +36 28 522 000, fax: +36 28 522 927, E-mail: Bernath.Gergely@mkk.szie.hu
INTRODUCTION
Eurasian perch (Perca fluviatilis) is a freshwater species with increasing interest in Europe. The artifical reproduction of perch is still being developed as the synchronization of spermiation and ovulation is very difficult. There is a little information available regarding the cryopreservation of perch sperm. Rodina et al. successed with cryopreservation in 2008, however, several important parameters need to be optimized. It is neccessary to improve the currently available protocols to expand our knowledge about artificial reproduction of this species. Thus, the objectives of our study were to carry our a systematic optimization of the parameters of cryopreservation of Eurasian perch sperm.
MATERIALS AND METHODS
Wild caught Eurasian perch (Perca fluviatilis) broodstock was used in the experiments. The males were not injected before the experiments. The females were injected with 500 IU per kg-1 of hCG and were incubated at 14 ºC before the fertilization experiment. Sperm was collected from anesthetized males using a catheter and stored at 4 ºC. Progressive motility was measured using a CASA system both before and after cryopreservation in every experiment. Lahnsteiner’s immobilizing and activating solutions were used to predilute and activate sperm (Lahnsteiner 2011).
Experiment 1. Selection of the best extender for cryopreservation: The effects of Tanaka extender (Szabó et al. 2005) and modified Lahnsteiner’s immobilizing solution (Lahnsteiner 2011) in combination with 10% methanol on the post-thaw motility of perch sperm from 7 males were investigated. Sperm was cryopreserved at a dilution ratio of 1:40 (v/v) in 0.5 ml straws in liquid nitrogen vapour. To measure the motility straws were thawed up to 13 seconds in a waterbath at 40 ºC.
Experiment 2. Selection of the best dilution ratio for cryopreservation: In this experiment sperm from 10 males was cryopreserved in modified Tanaka extender (Szabó et al. 2005) at dilution ratios of 1:1, 1:5, 1:10, 1:20, 1:30 and 1:40 (v/v). The cryopreservation and thawing process was identical to with that in Experiment 1.
Experiment 3. Effect of post-thaw storage on the quality of cryopreserved sperm: Experiment 3.1.
After thawing sperm of 10 males was stored for up to 1 hour at 4 ºC with motility measured every 15 minutes. Experiment 3.2. Sperm from 10 males was stored at 4 ºC for up to 6 hours after thawing. Motility was measured every 2 hours.
Experiment 4. Fertilization with cryopreserved sperm: For fertilization experiments gametes of 10 males and 1 female were stripped. The cryopreservation and thawing process was identical to that in previous experiments. After thawing eggs were immediately fertilized with all cryopreserved samples in duplicates. Fresh sperm was used for control fertilization in duplicates. Fertilized eggs were calculated with a stereomicroscope 3 days after fertilization.
RESULTS
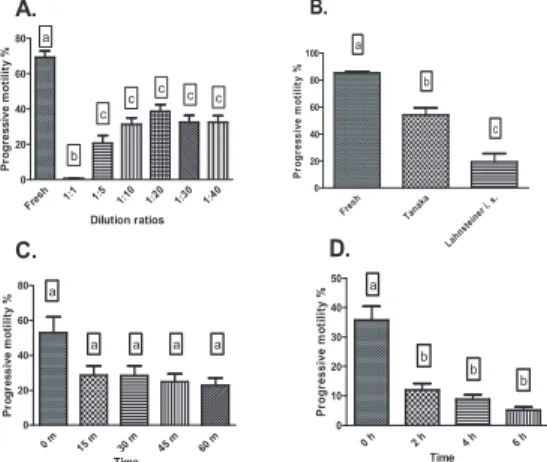
Cryopreserved sperm showed significantly higher motility with Tanaka extender (54,1±14 %) than with Lahnsteiner’s immobilizing solution (19,20±16,65%) (Figure 1B.). Motility of cryopreserved sperm was the highest (38,7±12,2 %) at the 1:20 sperm-extender and cryoprotectant ratio (Figure 1A.). In experiment 3.1. progressive motility reduced after 15 minutes, however it did not decrease for a further 60 min (Figure 1C.). In experiment 3.2. the results showed that the progressive motility
Session 2 Gamete Storage and Preservation
129 of cryopreserved sperm significantly reduced after 2 hours. From 2 to 6 hours the reduction of progressive motility was less apparent (Figure 1D.). In experiment 4. fertilization rate with cryopreserved sperm was 75,3 ±5,5 % whereas post-thaw motility was 58,48±24,5 %
Figure 1. Post-thaw motility of Eurasian perch sperm at various dilution ratios (A), with various extenders (B) and at different periods of post-thaw storage (C and D).
DISCUSSION AND CONCLUSIONS
We have found that the modified Tanaka extender originally described for the sperm of the European eel (Szabó et al. 2005) can successfully be applied to the cryopreservation of perch sperm. Contrary to previous findings (Rodina et al. 2008), where the recommended dilution ratio was 1:6, a dilution ratio of 1:20 was found to be optimal for cryopreservation of perch sperm.
Sperm of perch is sensitive to post-thaw storage as indicated by the sharp significant decrease of post-thaw motility after 2 hours. Thawed perch sperm is recommended to be used within 1 hour following thawing. Cryopreserved sperm was successfully used for the fertilization of perch eggs.
ACKNOWLEDGEMENTS
Research was supported by the TÁMOP 4.2.2/B-10/1-2010-011 “Development of a complex educational assistance/support system for talented students and prospective researchers at the Szent István University” project. Partially funded by COST Office (Food and Agriculture COST Action FA1205: AQUAGAMETE).
REFERENCES
Lahnsteiner, F., 2011: Spermatozoa of the teleost fish Perca fluviatilis (perch) have the ability to swim for more than two hours in saline solutions. Aquaculture 314, 221–224.
Rodina, M.; Policar, T.; Linhart, O.; Rougeot, C., 2008: Sperm motility and fertilizing ability of frozen spermatozoa of males (XY) and neomales (XX) of perch (Perca fluviatilis). J. App. Ichthyol. 24, 438–
442.
Szabó, G.; Müller, T.; Bercsényi, M.; Urbányi, B.; Kucska, B.; Horváth, Á., 2005: Cryopreservation of European eel (Anguilla anguilla) sperm using different extenders and cryoprotectants. Acta Biol. Hung.
56, 173–175.
Fresh 1:1 1:5
1:10 1:20 1:30 1:40 0
20 40 60 80
A.
a
b c
c c
c c
Dilution ratios
Progressive motility %
Fresh Tanaka
Lahnsteiner i. s. 0
20 40 60 80 100
B.
a b
c
Progressive motility %
0 h 2 h 4 h 6 h
0 10 20 30 40 50
D.
Time Progressive motility % a
b
b b
0 m 15 m
30m 45 m
60 m 0
20 40 60 80
C.
Time Progressive motility % a
a a
a a