ContentslistsavailableatScienceDirect
Journal of Pharmaceutical and Biomedical Analysis
jo u r n al ho me p a g e :w w w . e l s e v i e r . c o m / l o c a t e / j p b a
A corneal-PAMPA-based in silico model for predicting corneal permeability
Anna Vincze
a, Gerg ˝o Dargó
a, Anita Rácz
c,∗∗, György T. Balogh
a,b,∗aDepartmentofChemicalandEnvironmentalProcessEngineering,BudapestUniversityofTechnologyandEconomics,M ˝uegyetemRakpart3.,1111, Budapest,Hungary
bDepartmentofPharmacodynamicsandBiopharmacy,UniversityofSzeged,Eötvösu.6.,6720,Szeged,Hungary
cInstituteofMaterialsandEnvironmentalChemistry,ResearchCentreforNaturalSciences,MagyarTudósokKrt.2.,1117,Budapest,Hungary
a rt i c l e i nf o
Articlehistory:
Received3February2021
Receivedinrevisedform14June2021 Accepted15June2021
Availableonline17June2021
Keywords:
Cornealpermeability Invitro
Non-cell-basedmodel PAMPA
Insilicomodel QSPR
Quantitativestructure-property relationships
Lipophilicity Polarsurfacearea
a b s t ra c t
Thecapabilitytopredictcornealpermeabilitybasedonphysicochemicalparametershasalwaysbeen adesirableobjectiveofophthalmicdrugdevelopment.However,previousworkhasbeenlimitedto caseswhereeitherthediversityofcompoundsusedwaslackingortheperformanceofthemodels waspoor.Ourstudyprovidesextensivequantitativestructure-propertyrelationship(QSPR)modelsfor cornealpermeabilitypredictions.Themodelsinvolvedinvitrocornealpermeabilitymeasurementsof189 diversecompounds.Preliminaryanalysisofdatashowedthatthereisnosignificantcorrelationbetween corneal-PAMPA(ParallelArtificialMembranePermeabilityAssay)permeabilityvaluesandotherpharma- cokineticallyrelevantinsilicodrugtransportparameterslikeCaco-2,jejunalpermeabilityandblood-brain partitioncoefficient(logBB).TwodifferentQSPRmodelsweredeveloped:oneforcornealpermeability andoneforcornealmembraneretention,basedonexperimentalcorneal-PAMPApermeabilitydata.Par- tialleastsquaresregressionwasappliedforproducingthemodels,whichcontainedclassicalmolecular descriptorsandECFPfingerprintsincombination.Acomplexvalidationprotocol(includinginternaland externalvalidation)wascarriedouttoproviderobustandappropriatepredictionsforthepermeability andmembraneretentionvalues.BothmodelshadanoverallfitofR2>0.90,includingR2-valuesnotlower than0.85forvalidationruns,andprovidequickandaccuratepredictionsofcornealpermeabilityvalues foradiversesetofcompounds.
©2021TheAuthor(s).PublishedbyElsevierB.V.ThisisanopenaccessarticleundertheCCBYlicense (http://creativecommons.org/licenses/by/4.0/).
Abbreviations: ADME,absorption,distribution,metabolism,excretion;API,activepharmaceuticalingredient;Caco-2,celllineofheterogeneoushumanepithelialcol- orectaladenocarcinomacells;clogD7.4,logofcalculateddistributioncoefficientatpH7.4;clogP,logofcalculatedpartitioncoefficient;clogPeCaco-2,logofcalculatedCaco-2 permeability;clogPejejunal,logofcalculatedjejunalpermeability;DOOD,D-optimaloniondesignalgorithm;fbu,fractionunboundinthebrain;GA,geneticalgorithm;HBD, numberofhydrogenbonddonors;HPLC,highperformanceliquidchromatography;ka,absorptionrateconstant;logBB,blood-brainpartitioncoefficient;logD7.4,logof distributioncoefficientatpH7.4;logDI7,65,degreeofionizationatpH7.65;logP,logofpartitioncoefficient;logPe,logofeffectivepermeability;logS,logaqueoussolubil- itymeasuredinmol/L;MR,membraneretention;MW,molecularweight;NN,neuralnetworks;PAMPA,parallelartificialmembranepermeabilityassay;PBS,phosphate bufferedsaline;PC,phosphatidylcholine;PCA,principalcomponentanalysis;P-gp,P,-glycoprotein;PLS,partialleastsquares;PLS,COMP.numberofPLScomponentsinthe model;PPB,plasmaproteinbinding;PSA,polarsurfacearea;Q2CV,goodnessoffitforthecross-validation;R2P,goodnessoffitfortheexternalvalidation;QSAR,quantitative structure-activityrelationship;QSPR,quantitativestructure-propertyrelationship;R2,goodnessoffitforthecalibration;RMSEC,rootmeansquarederrorofcalibration;
RMSECV,rootmeansquarederrorofcross-validation;RMSEP,rootmeansquarederrorofexternalvalidation;TPSA,topologicalpolarsurfacearea;VIP,variableimportance projection;max,thewavelengthofmaximumabsorbance;Y-SCR,CVgoodnessoffitforthecrossvalidationusingYscrambling;Y-SCRP.,goodnessoffitforthetestprediction usingY-scrambling.
∗Correspondingauthorat:DepartmentofChemicalandEnvironmentalProcessEngineering,BudapestUniversityofTechnologyandEconomics,M ˝uegyetemrakpart3., 1111,Budapest,Hungary.
∗∗Correspondingauthor.
E-mailaddresses:racz.anita@ttk.hu(A.Rácz),balogh.gyorgy@vbk.bme.hu(G.T.Balogh).
https://doi.org/10.1016/j.jpba.2021.114218
0731-7085/©2021TheAuthor(s).PublishedbyElsevierB.V.ThisisanopenaccessarticleundertheCCBYlicense(http://creativecommons.org/licenses/by/4.0/).
1. Introduction
Theeyeisachallengingtargetoftherapy.Becauseofitscompli- catedanatomicstructureandsmallabsorptivesurface,itisdifficult to achievetherapeutic druglevelsin theinterior oftheeye by topical dosageforms (eye drops, ointments,in-situgel-forming systems,etc.).Moreinvasiveonessuchasintraocularinjections orimplantscanbeveryusefulformaintaininghighdruglevelsin both theanteriorandposteriorsegment[1–3].Topicaladminis- trationisthemostcommonwayofanteriorsegmenttreatment asitisnon-invasive,easyandverycomfortable.However,ithas manydrawbacksincludinglowbioavailabilitydespitethefactthat the activepharmaceuticalingredient (API) is usuallyapplied in highdoses[4].Blinking,tearformationandnasolacrimaldrainage causeahugedropindruglevelasthesolutionsarewashedaway withinthefirst15–30secondsfromtheeyesurface [5,6]. Typi- callynomorethan5%bioavailabilitycanbereachedintheanterior segmentbytopicaladministrationduetothesedefensemecha- nisms,andmembranepenetrationissues[4–7].Byusingdosage formswithhighviscosityandmucoadhesivitytheresidencetimeof drugscanbeincreased,however,theseointmentsandinsitugelling systemsoftencauseirritationandblurredvision[1,4,5,8].Higher druglevelscan bereachedin theposteriorsegmentof theeye bysystemicadministrationorintraocularinjectionsandimplants [2,3,5,9].Whilethefirstmayresultinsystemicsideeffectsdueto highdosagethelatterdoesnotrequirefrequentuse,although,it maycauseendophthalmitis,lens damageorretinaldetachment [4,10,11].Despitelow patientcompliance,theintraocularinjec- tions(usuallytargetingthevitreous)arestillthemostcommonly usedposteriorsegmenttherapiessincetherearenootherefficient deliveryroutesforreachinghighdruglevelsintheposteriorwith minimalsystemicsideeffects[9].
The possible routes of absorption are the corneal and non- corneal pathwaysinterms oftopicaladministration [5,6,10].In thecase ofnon-cornealroutesthedrugpenetratesthroughthe conjunctivaand/orsclerawhich isnotveryeffectiveduetothe presenceoflocalcapillarybedsthatcontinuouslyremovethedrug, transferring itintothesystemic circulation.On theotherhand, thecornealrouterepresentsthemainabsorptionpathformost ophthalmictherapeutics(small,lipophiliccompounds)andforthis reason,itisthemoststudiedareabothintermsofdrugdiscovery anddrugformuladevelopment[4,5].Thecorneaisaclearandavas- culartissueconsistingof5layers:cornealepithelium,Bowman’s layer,stroma,Descemet’smembraneandendothelium.Although therelativethicknessesofcornealepithelium,stromaandendothe- liumareabout0.1:1:0.01,thethinnestcornealepitheliumisthe mainbarrierofdrugabsorptionintotheeye[7,10].
Likeinthecaseofothertopicaldosageforms,theinvivohuman pharmacokineticcharacterizationofAPIsinophthalmictreatment isgreatlyhamperedbythedifficultyofdirectlyexaminingtopical drugconcentrations.Furthermore,duetotechnicaldifficultiesno suitableinvivoanimaltestingsolutionisavailableeither[12].For preclinicalophthalmicabsorptionstudiesexvivoandinvitrocel- lularmodelsareavailable.Forexvivostudiesexcisedrabbitcornea isusedmostcommonlybecauseofeasyaccessandanatomicsim- ilaritiesbetweenrabbitandhumaneyes[10,13].However,there arealsosomedifferencesbetweenthetwospeciesandthusthe penetrationisgenerallymuchhigherandcannotbecorrelatedade- quatelytohumans[10].Invitrostudiesapplyrabbitorhumancell cultureswhichareconsideredasamoreethicalalternative.Inthis case,mostlycornealepithelialcellsareusedconsideringthemain roleofcornealepitheliumindrugtransport,however,itisdifficult toisolateoculartissuesforthispurposeandthelifespanofthese primarycellsisveryshort[13,14].Althoughimmortalizedcelllines provideaninexhaustiblesupplyofcellstheymayexhibitabnormal geneexpressionorbiologicalfunctionsespeciallyafterseveraldivi-
sions.Also,penetrationacrosscellculturesisonlyacrudeestimate atbestsinceasingletypeofcellcannotrepresentthecomplexity ofthecornealbarrierasawhole[13].
Thecapabilitytopredictcornealpermeabilitybasedonphysic- ochemical parameters hasalwaysbeena desirable objective of ophthalmicdrugdevelopment.Forthispurpose,greateffortshave beenmadetodeveloppredictivemodelsstartingwiththeworkof SchoenwaldandWard[15].Asummaryoftheearlydevelopment stagescanbefoundinTable1.Thesemodelswerebasedonjust afewphysicochemicalpropertiesbutweretypicallynotgenerally applicable–theirdiversityandperformancewerelimitedtoonly certaingroupsofmolecules.
Inthepastfewdecades,quantitativestructure-propertyrela- tionship (QSPR) techniques have become a well-established methodforcorrelatingmolecularstructureandphysicochemical propertiesofchemicalcompoundswiththeuseofseveraldescrip- torsandnowadaystheyarecommonlyusedforthefastprediction of differentADME(absorption, distribution,metabolism, excre- tion)relatedmolecularproperties[21,22].Therehavebeenafew attemptstodevelopQSPRmodelsalsoforthepredictionofcorneal permeability.Kidronetal.usedpartialleastsquares(PLS)regres- sionfor 69 non-congenericcompoundsand concluded thatthe totalnumberofputativehydrogenbonds(HBTOT)andlogarithmic distributioncoefficients(logDpH7.4,logDpH8.0)werethemostsig- nificantdescriptorsforcornealpermeability[12].Thesignificance oflipophilicityin cornealpenetrationhasbeenrecognizedlong ago,whileforionizablecompounds(mostoftheophthalmicdrugs) thedistributioncoefficientmeasuredatthepHofaqueoushumor andtearfilm(pH7.2–7.6) hasbeenfoundtobeamoreproper descriptorintermsofmembranepermeability[5,8].Inthework ofGhorbanzadeetal.thesamedatasetwasanalyzedbystepwise multivariatelinearregression (MLR),andneuralnetworks(NN) aswell[23].Withthemultilayerperceptron-NNtechnique,they establishedanonlinearmodelforcornealpermeabilitywithmod- erateperformance. AlthoughKidronandGhorbanzade analyzed morecompoundscomparedtotheirpredecessors,thedatacame fromseveralsourceswithdifferentexperimentalconditions.
In2018,Ramsayetal.producedacornealandconjunctivalQSPR modelbyPLSandprincipalcomponentanalysis(PCA)basedonan exvivodatasetof32smallmolecules.Theyfoundthatcornealper- meabilityhasaninverserelationshipwiththepolarsurfacearea (PSA)andthenumberofhydrogenbonddonors(HBD)[24].How- ever,thepredictivepoweroftheirmodelwasrelativelymodest.
Uptothepresentday,therehavebeenseveralmodelsdeveloped forpredictingcornealpermeability, butthere isstillnoreliable QSPRmodelbasedonalargeandhomogeneousdataset.Ofcourse, itisnotaneasymissionsinceexvivoexperimentshaveseriouscon- straintslikethedemandfornumerousanimals,appropriatetissue viabilityandhighcosts.Invitrocellculturemodelsarebetterin termsofcost-effectivenessandthroughput,however,theuseof cell-basedassaysisnotarobustprocess.
To solvethis problem, we have recently developed a novel invitronon-cellularPAMPA-basedmethodforinvestigatingthe cornealpermeabilityofAPIs[25].Corneal-PAMPAisarobust,high- throughputassayusingeasilyaccessiblematerialslikephosphate bufferedsaline(PBS),phosphatidylcholine,dodecane,hexaneand chloroform.Fordevelopingthemethod,exvivorabbitcornealper- meabilitydatawereusedfromheterogeneousdatasetsinterms ofexperimentalconditions.Despitetheuseofheterogeneousdata, thePAMPAmodelwithstandardizedexperimentalconditionspro- videdhighlyreproduciblepermeabilityvaluesthatcorrelatedwell withtheexvivodata(R2=0.88).Thecorneal-PAMPAmethodology canprovidemanycornealpermeabilityvaluesasahomogeneous datasetinashorttimeperiodandthereforeitisasuitablebasisfor thedevelopmentofaninsilicomodel.
Table1
Detailedsummaryoftheearlymodelsforcornealpermeabilitydetermination.
Equation Typeofmolecules(andnumbers) Molecularproperties Ref.
parabolic steroids(11) logP [15]
parabolic N-alkyl-p-aminobenzoateesters(6) logP [16]
parabolic -blockingagents(12) logP,logDpH7.65,logMW,logDI7.65* [17]
parabolic/sigmoidal -blockingagents(13) logP [18]
parabolic -blockers,steroids,miscellaneouscompounds(32) logD,logP(logPoctanol-logPalkane) [19]
MLRmodel -blockers,steroids,miscellaneouscompounds(30) quantumchemicaldescriptors(3) [20]
*DegreeofionizationatpH7.65.
In the present work, the corneal permeability of 189 APIs selected froma diversechemical spacewasdetermined bythe corneal-PAMPA method [25] experimentally and used to build a robustQSPRmodelfor predictingcornealpermeability,which couldbeausefultool inthedevelopmentoffutureophthalmic agents.
2. Materialsandmethods 2.1. Materials
Analytical grade solvents like acetonitrile, chloroform, hex- ane,dodecaneandformicacidwerepurchasedfromMerckKGaA (Darmstadt,Germany).Phosphatebufferedsaline(PBS)powder,l-
␣-phosphatidylcholine(PC)andallinvestigatedcompounds(See TableA1.,AppendixA)werepurchasedfromSigmaAldrichCo.Ltd.
(Budapest,Hungary)exceptforbetaxololhydrochloride,bevantolol hydrochloride, bromfenac sodium, bufuralol hydrochloride, 11- deoxycorticosterone,ethoxzolamide,flurbiprofen,nepafenacand penbutolol,whichwerepurchasedfromTorontoResearchChemi- calsInc.(NorthYork,Toronto,Canada).Distilledwaterwaspurified bytheMilliporeMilli-Q®140GradientWaterPurificationSystem.
2.2. Invitropermeabilitystudies
Thecorneal-PAMPAmeasurementswerebasedonourprevi- ousreport[25].BrieflyallcompoundsweredissolvedinPBSbuffer (pH7.4)tomakesolutionswith100Mnominalconcentration.
DuetothenegativeeffectofusingDMSOasacosolventonthe goodnessofpredictioninthecorneal-PAMPAassaysystem(accord- ingtoourpreviousreport[25]),noDMSOwasusedforthisstudy either.300LoftheinitialPBSsolutionswereplacedintoa96- wellpolypropyleneplate(A)(Agilent,Waldbronn,Germany).For thecorneal-PAMPAmembrane,16mgPCwasdissolvedinasol- ventmixtureofchloroform,hexaneanddodecane5:70:25%(v/v) in atotal volumeof600L.Eachwelloftheacceptorplate(B) (MultiscreenAcceptorPlate,MSSACCEPTOR;Millipore)contained 300LPBSandeachwellofthedonorplate(MultiscreenTM-IP, MAIPN4510,poresize0.45m;Millipore)(C)wascoatedwith5L ofthelipidsolution.Afterevaporationofhexaneandchloroform, thedonorplatewasinsertedintotheacceptorplate,andeachwell ofthedonorplatewasfilledwith150LPBSsolutionsofcom- poundsfromplateA.Afterthat,thedonorplatewascoveredwitha sheetofwettissuepaperandplatelidtopreventevaporationofthe solventduringthe4hincubation(35◦C,HeidolphTitramax1000).
Ineachassay,tworeferencecompoundsweremeasured:buspirone andhydrocortisonewithhighandlowcornealpermeabilityvalues, respectively.
For HPLCanalysisatleast120Laliquotsweretakenout of everywellofplatesBandCandtransferredto96-wellpolypropy- lene platesandsealed.TheinitialplateAwasanalyzedaswell.
Theeffectivepermeabilityandmembraneretentionofdrugswere calculatedusingthefollowingequations[26]:
Pe= −2.303
A×(t−ss)×
11+rv
×lg
−rv+
1+rv 1−MR
× cD(t) cD(0)
(1) wherePe istheeffectivepermeability (cm/s),Aisthefilterarea (0.3cm2),tistheincubationtimeofPAMPAassay(14400s),ssis thetimetoreachsteady-state(s),rvisthevolumeratioofaqueous compartments(VD/VA),VDandVAarethevolumesinthedonor (0.15cm3)andacceptorphase(0.3cm3),cD(t)istheconcentration ofthecompoundafterincubationonplateC(mol/cm3),cD(0)isthe concentrationofthecompoundonplateA(mol/cm3)andMRisthe membraneretentionfactor,definedas:
MR=1− cD(t)
cD(0)− VAcA(t)
VDcD(t) (2)
wherecA(t)istheconcentrationofthecompoundafterincubation onplateB(mol/cm3).Intablesandfiguresweusuallyusethisterm as100*MR(%).
2.3. HPLCanalysis
Quantitativechromatographicanalyseswerecarriedoutusing anAgilent1260liquidchromatographysystem(AgilentTechnolo- gies,PaloAlto,CA,USA)onaKinetex®2.6mC18100ÅLCcolumn (30×3mm)withamobilephaseflowrateof1.1mL/min.Mobile phaseAandBconsistedof0.1%(v/v)formicacidinwater,andace- tonitrile/water(95/5)with0.1%(v/v)formicacid,respectively.A 3.91minlonglineargradientprogramwasused:inthefirst0.3min 0%B,between0.3and1.8min0–100%B,then100%Bwaskept for0.6minand finally,at2.41the100%Bwasdroppedto0%.
Thiswasfollowedbyanequilibrationperiodof1.5minpriorto thenextinjection.Theinjectionvolumewas6L.Chromatograms wererecordedbyadiodearraydetectorinthewavelengthrange of200–500nm,andintegrationwascarriedoutatthemaxofeach compound.Data acquisitionand analysiswerecarriedoutwith ChemStationB.04.03.
2.4. Datasetgeneration
Theapplieddatasetcontainedtheexperimentalcorneal-PAMPA permeabilityandthemembraneretentionvaluesof189APIsbased onthepreviouslymentionedexperimentalmodel[15].Thedom- inantprotonationstateatpH7.4wasassignedtoeachcompound withtheChemAxonCalculator(cxcalc)[27]andSchrödinger(Lig- Prep)[28,29].Consequently,theappropriate3Dstructureswere usedforthemoleculardescriptorgeneration.
Classical1,2and3Dmoleculardescriptorsandextendedcon- nectivityfingerprints(ECFP)weregeneratedfortheAPIswiththe DRAGON7.0software[30].Thedefinitionoftheappliedmolecular descriptorscanbefoundintheworkofTodeschinietal.[31].More- over,anadditional80physicochemicalpropertieswerecalculated bytheACD/Perceptasoftware[32].Aninter-correlationlimit of 0.997wasusedforthefilteringoftheclassicaldescriptors[33].Con- stantdescriptorswerealsoexcludedfromthedataset.FortheECFP
fingerprints,defaultparameterswereappliedinDRAGON7.0,with amaximumradiusof4andafingerprintlengthof1024[34].The finalnumberofgeneratedmoleculardescriptorswas3302,which werefurtherusedfortheQSPRmodelbuilding.
2.5. QSPRmodeling
Partialleastsquares(PLS)regressionwasappliedasaclassical andfrequentlyusedregressionalgorithmforthemodelbuilding.
InPLSregression,latentvariablesarecalculated,whichcanbeused for the predictionof theY dependentvariable,in ourcase the corneal-PAMPApermeabilityvaluesandthemembraneretention.
Asawell-establishedtoolinQSAR/QSPRanalysis,thereisnoneed toexplainthePLSalgorithminmoredetailotherthanreferringto thetutorialpaperofGeladiandKowalski[35].Theoptimalnum- berofPLScomponentsforthemodelswasselectedbasedonthe globalminimumoftherootmeansquarederrorofcross-validation (RMSECV).Outlierselectionofthesampleshasbeencarriedoutby theplotofthefirsttwolatentvariables(PLScomponents).The95
%confidenceellipsewasusedforthedeterminationoftheoutliers.
TherangeoftheYvariable(corneal-PAMPAvalues)wasalsomod- ifiedto0−50×10−6cm/s,duetothepoorcoverageoutsideofthis range.
In eachmultivariatecalibrationwiththousandsofmolecular descriptors, the use of variable selection techniques [36] is an essentialstepfortheappropriatemodelbuildingwithgoodpredic- tionperformance.Inourcase,thevariableimportanceprojection (VIP)andgeneticalgorithm(GA)methodswerecombinedforthis purpose.First,thenumberofvariableswasdecreasedtothemost important20%basedonVIPvalues,thenageneticalgorithmwas usedontheremainingvariables.Thisformofpre-filteringwithVIP scoreswasnecessaryinordertodecreasethecalculationtimefor themoretime-demandingGAprotocol.Themajorparametersof thegeneticalgorithmwerethefollowing:thepopulationsizewas 64,thepercentageoftheinitialtermswas30,thealgorithmwas runforahundredgenerationsanddoublecross-overwasused.
ModelbuildingwascarriedoutinPLSToolbox[37].Validation ofthemodelsisindispensableforreliableprediction,thereforeour modelswerevalidatedinthreedifferentways:cross-validation, externalvalidationandY-scrambling[38].Trainingandtestsplits werecarriedoutbytheD-optimaloniondesignalgorithm(DOOD) implemented inPLStoolbox[39].Thedatasplit ratiowas80% Training–20 %Testset.Thetraining sampleswereusedfor7- foldcross-validation,whichmeansthat1/7ofthesampleswere predictedbasedontheother6/7ofthesamples,andthisprocess wasrepeatedwhileallthepartitionshavebeenpredictedexactly once.Thefoldsofsampleswereselectedrandomly,thustwenty iterationswereimplementedintotheprotocol.Seven-foldcross- validationisawell-knownoptionforcross-validationprocesses.
Theoptimalnumberoffoldsshouldbebetween5and10basedon theworkofHastieetal.[40].Thetestsplitsofthesampleswere usedfortheexternalvalidation,wheretheYtargetvaluesofthe moleculeswerepredictedbasedonthecalibrationmodel.Finally, Y-scrambling (randomlyreorderedYvector)canverifythatour modelsarefarbetterthantheuseofrandomnumbers.Thiscan helpustocheck“chancecorrelations”andwhetherthemodelsare overfittedornot.Anadditionalmethod,calledthepermutationtest wasalsoappliedtotestthesignificanceofthedifferencebetween theactualandtherandommodel.Herethemainhypothesisisthat ouroriginalmodelisnotsignificantlydifferentfromtherandomly reorderedYvectorones.Wilcoxon,SignandrandomizationT-tests wereappliedfortesting.
PerformanceparameterssuchasR2(calibration),Q2CV(cross- validation),R2P(externalvalidation)andQ2Fn[41],therootmean squared error of calibration, cross-validation and external vali- dation(RMSEC/RMSECV/RMSEPrespectively),themeanabsolute
error(forcalibration,cross-validationandexternalvalidation)the coefficientofconcordance(CCC,likewiseforallcalibrationandval- idationsteps)[42]etc.werecalculatedtoappropriatelydetermine thequalityofthemodels.
3. Resultsanddiscussion 3.1. PreliminaryanalysisoftheAPIs
Inourpreviouswork,weinvestigatedtherelationshipsbetween corneal-PAMPApermeability(logPe)andmembraneretentionval- ues(MR) incomparisonwithexperimentalCaco-2 permeability dataandcalculatedphysicochemicalproperties,suchasmolecular weight(MW),topologicalpolarsurfacearea(TPSA)andlipophilic- itydescriptors(logP,logDpH7.4),inthecaseof50structurallyand physicochemicallydiversecompounds[43].Duetoweakcorrela- tions(R2=0.018−0.506)weconcludedthatcornealpermeability cannotbepredictedbasedononlythesephysicochemicaldescrip- torsandforthisreason,alargermeasuredpermeabilitydataset is needed to be able to carry out a QSPR analysis. As part of ourconclusion,weestablishedthattheweakcorrelationbetween corneal-PAMPApermeabilityandCaco-2(gastrointestinal)perme- ability(R2=0.125)confirmsthatourmodelisindependentofthe generallyacceptedgastrointestinalpermeabilitymodelandmay supportitsadequacyforcornea-specificinvitromeasurements.
Inthecurrentwork,189APIs(fromwhich50APIshavealready beenmeasuredinthepreviouswork[43])wereselectedfroma diversechemicalspaceandtestedinthecorneal-PAMPAmodelto producedatasetforinsilicomodeling.(Seeexperimentaldatain TableA1.,AppendixA)Beforecomputationalmodeling,weeval- uatedthemaincharacteristicsofthecompoundsbasedontheir physicochemical properties like logP, logDpH7.4,MW, TPSA and acid-basecharacteristics(Fig.1).
Weinvestigatedtherelationshipbetweencorneal-PAMPAper- meabilityandtheabove-mentionedphysicochemicalparameters (Fig.2).Forlinearregressionanalysis,theGraphPadPrismsoftware wasused[45].Theresultprovedtobealikeaseachoftheseparam- etersshowedweakcorrelationwithcornealpermeability,onlythe followingtendenciescouldbeobserved:thecornealpermeability decreasedwithincreasingTPSAwhileitincreasedwithincreasing logPandlogDpH7.4values.However,themolecularweightseemed tohavenoeffectoncornealpermeability.Theneutralandbasic compoundsexhibitedhigherpermeabilityvaluesthantheacidic oramphotericones,which observationcorresponds tothegen- eraltrendformembranepermeabilityofdrugsitashasalsobeen reportedpreviously[46].Thebestsquaredcorrelationcoefficient (R2=0.356)wasobservedinthecaseoflogDpH7.4,whichisprobably duetothefactthatthemeasurementswerecarriedoutatiso-pH 7.4conditions.
The relationship between membrane retention and these physicochemical parameters hasalso been investigated. In the case ofTPSAand logPonlytrends couldbeobserved withlow squaredcorrelationcoefficients(R2=0.289and0.367respectively).
TheMRvaluesincreasedwithincreasinglogPanddecreasedwith increasingTPSA,whichisinaccordancewiththefactthatthemore lipophilic(andlesspolar)theAPIis,themorelikelyitgetsintothe membrane.Theanalysisresultedinverypoorfitswiththeother twoparameters.(SeeFigureB1.,AppendixB).Thecorneal-PAMPA permeabilityvalueswerealsocomparedwithotherpermeability- relatedinsilicovalueslikeCaco-2permeability(pH7.4,at350rpm stirringrate)jejunalpermeability[47]and blood-brainpartition coefficient[48](logBB),whichwerepredictedbytheACD/Percepta software[32]asagoldstandardtoolindrugdiscovery(Fig.3).
The Caco-2 model applies human epithelial colorectal ade- nocarcinoma cells thus, unlike the PAMPA model, it contains
Fig.1.Histogramsofphysicochemicalpropertiesof189compounds.Physicochemicalpropertiesincludemolecularweight,topologicalpolarsurfacearea,calculated logpartitioncoefficient,calculatedlogdistributioncoefficientatpH7.4,andacid-basecharacteristics.TheTPSA,clogPandclogDpH7.4valueswerecalculatedwiththe ACD/Perceptasoftware[32]andtheacid-basecharacteristicsweredeterminedwiththeChemAxon/MarvinSketchsoftware[44].
activetransporters.Forcomparingthecorneal-PAMPAandCaco- 2permeabilityvalues,9APIs(hydrocortisone,emetine,etoposide, loperamide,quinine,risperidone,sparfloxacin,trimethoprim,ver- apamil)werelabeledwithredstarsonFig.3,whicharepotential substrates of P-gp efflux pump according to the prediction of ACD/Perceptasoftware[32].Forthepropercomparisonbetween thedifferentassays,compoundsthatareknowntobepenetrating viaactivetransportshouldbeavoided.Toclarifythedifferences intheirpenetrationbehavior,squaredcorrelationcoefficientsare representedhere,whichrefertothedatasetswithout(R2)andwith (R2*)theP-gpsubstratesaidedbyactivetransport.Similarly,to ourformer conclusion, onlya weakcorrelationcouldbedeter- minedbetweencorneal-PAMPAandCaco-2permeabilityvalues.
Aweakcorrelationwasfoundinthecaseofjejunalpermeability andblood-brainpartitioncoefficienttoo,whichindicatesthatthe corneal-PAMPAisnotauniversalpermeabilitymodelandthatthe trans-cornealdiffusionprocessispresumablygovernedbydiffer- entfactorsthanintestinalorblood-brain-barrierpenetration.Thus, theresultsofthepreliminaryanalysisclearlyverifiedtheneedfor auniqueQSPRmodelforcorneal-PAMPApredictions.
3.2. Corneal-PAMPApermeabilityQSPRprediction
Inthemodel-buildingphase,theoriginalmatrixcontained3302 molecular descriptors(including the predictedphysicochemical ones,classical1,2and3DdescriptorsandtheECFPfingerprint)as Xvariables,andcorneal-PAMPAexperimentalresultsastheYvec- tor.ThetotalnumberofAPIswherecorneal-PAMPAexperimental values couldbedeterminedwas151.Outlierselectionwasper- formedonthedataset:a)firsttheAPIsoutsideofthewell-covered
0−50×10−6cm/srangewereexcluded,b)thenthefirsttwoPLS componentswereplottedagainsteachotherand95%confidence ellipsewascalculatedtoexcludetheoutliersbasedonthemolec- ulardescriptors.Inthefinalform,143APIswereincludedinthe dataset.
Inthenextstep,thematrixwassplitintotrainingandtestset, whichwasobligatorytocarryoutapropervalidationofthemodel.
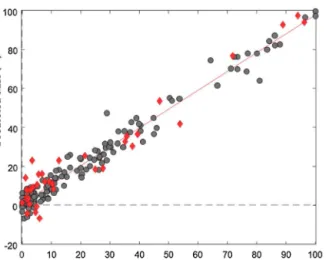
WiththeDOODalgorithm,80%ofthecompoundswereselected forthetrainingset.Thus,114APIswereappliedforthecalibration andcross-validationof themodel.Autoscaling(standardization) wasusedasdatapretreatment.Wehaveselected100molecular descriptorswiththecombinationofVIPscoringandgeneticalgo- rithmfromtheoriginaldescriptorset.Inthe7-foldcross-validation part,RMSECVvalueswereusedforthedeterminationofthenum- berofPLScomponents.TheglobalminimumoftheRMSECVvalues wasatfivePLScomponents.Cross-validationwasperformedwith arandomizedselectionin20iterations.Thefinalcalibrationcurve alongwiththetestvalidationsamplesbasedon5PLScomponents isshowninFig.4ab.
Thesampleswerewell-fittedtothecalibrationcurveandthe errorswerehomogenousalongtherange.Itisclearlyseeninthe performancesaswell:theR2(goodnessoffitforthecalibration)and Q2CV(goodnessoffitforthecross-validation)were0.953and0.866, respectively.Theperformanceofthetestvalidationwas0.862.The remainingperformanceparametersofthemodelaresummarized inTable2.
Outoftheselected100variables,themostimportantphysic- ochemical descriptors (based on their VIP scores) were the following: absorption rate constant (ka), jejunal permeabil- ity (clogPejejunal,pH=7.4), fraction unbound in the brain (fbu),
Fig.2. PAMPA-relatedcornealpermeabilityasafunctionofphysicochemicalproperties.Correlationbetweencornealpermeabilityandmolecularweight(a.),topological polarsurfacearea(b.),calculatedlogpartitioncoefficient(c.),calculatedlogdistributioncoefficientatpH7.4(d.)andacid-basecharacteristics(e.)ofthecompoundswiththe squaredcorrelationcoefficient(R2).TPSA,clogPandclogDpH7.4valueswerecalculatedwiththeACD/Perceptasoftware[32]andtheacid-basecharacteristicsweredetermined bytheChemAxon/MarvinSketchsoftware[44].ThecorrelationcoefficientsweredeterminedusingtheGraphPadPrismsoftware[45].Thebluedottedlinesstandforthe95
%confidencebands.
clogDpH7.4,plasmaproteinbinding(PPB).Allthesedescriptorshad VIP(variableimportance)scoresabove1.3.Theresultscorrespond to thekinetic behavior of the permeability parameteras these descriptorsarerelatedtopassivetransportofdrugs.Inaddition, the fact that predicted data of neither thegastrointestinal (ka, clogPejejunal),northeblood-brainbarriertransport(fbu)insilico modelscoulddescribethecorneal-PAMPA permeabilityprocess, suggeststhecomplexityofourmodel,whichisalsosupportedby ourconclusionsinthepreliminaryanalysis.Ionizationstateand lipophilicityarealsowell-knowndeterminantsofthepermeabil- ity process [46,49,50], which are alsopresent herein theform ofclogDpH7.4.Additionally,linearregressionanalysiswascarried outinthecaseofka,PPBandfbu againstlogPe andonlyaweak correlationwasfoundineachcase,whichsupportsourprevious observationthatusinglinearregressionofonlyoneparameterata timecouldnotresultinagoodmodelforprediction(SeeFigureB2., AppendixBandTableA2.,AppendixA).
Yscramblingwasalsousedforthevalidationofthemodeland tocheckthepossibilityofoverfitting.Theexperimentalcorneal- PAMPAvalueswererandomizedinthedatasetforthisvalidation.
Thepredictionperformancesat5PLScomponentsweredecreased properlyto0.016and0.14forcross-validationandtestvalidation, respectively.Thepermutationtest(with50iterations)hasfounda significantdifferencebetweenourfinalmodelandtherandomized one(at␣=0.05level).FortheresultofthepermutationseeTable A3,AppendixA.
3.3. Corneal-PAMPAmembraneretentionQSPRprediction
Inthecaseofmembraneretentionprediction,thesameproto- col(withthesameparameters)wascarriedoutasinthecaseof permeabilityprediction.Theselectedrangeofmembrane reten- tionwasbetween0and100%.Aftertheoutlierselectionprocess, 180APIsoutof189wereincludedinthedataset.Theremaining
Fig.3.PAMPA-relatedcornealpermeabilityasafunctionofvariousinsilicotransportparameters.CorrelationbetweencornealpermeabilityandcalculatedCaco-2 permeability(pH7.4,at350rpmstirringrate)(a.),calculatedjejunalpermeability(b.)[47]andcalculatedblood-brainpartitioncoefficient(c.)[48]ofthecompounds withsquaredcorrelationcoefficients(R2).InthecaseofCaco-2permeability(a.)recalculatedsquaredcorrelationcoefficient(R2*)wasalsointroducedincludingnineP-gp substrates.clogPeCaco-2,clogPejejunalandclogBBvalueswerecalculatedwiththeACD/Perceptasoftware[32].ThecorrelationcoefficientsweredeterminedusingtheGraphPad Prismsoftware[45].Thebluedottedlinesstandforthe95%confidencebands.
Fig.4. ab.ThePLScalibrationcurve.a)Predictedcorneal-PAMPAvaluesareplottedagainsttheexperimentalones.InFig.4b)themodelistransformedtologarithmic scale.Calibrationsamplesaremarkedwithgreycircles,whiletestvalidationcompoundsaremarkedwithreddiamonds.
ninecompoundswereexcludedduetotheirnegativemembrane retentionvalues(theywereoutofthe0–100%range).Intotal,83 moleculardescriptorswereincludedinthematrixafterthevari- ableselectionprocess(VIPscoresandgeneticalgorithm).Classical physicochemical,two-dimensional,three-dimensionaldescriptors andfingerprintbitpositionswereincludedintheselectedsetof variables.ThesameDOODalgorithmwasusedforthetraining-test split.Thetrainingsetcontained144compounds(80%),whilethe remaining36compoundswereusedfortestvalidation.Seven-fold cross-validationwasusedfortheinternalvalidationofthemodel with20iterations,andeightPLScomponentswereappliedbased
ontheglobalminimumoftheRMSECVcurve.Thefinalcalibration modelwiththecalibrationandtestsamplestogetherisplottedin Fig.5.Thesampleswerewell-fittedtothecalibrationcurve(even thetestones)andtheerrorswerehomogeneousalongtherange.
TheR2 valueofthecalibrationwas0.97,whiletheQ2CVandQ2 predictionwere0.91and0.94,respectively.Alltheperformance parametersaresummarizedinTable2.
Themostimportantphysicochemicalpropertiesamongstthe83 selecteddescriptors(basedontheirVIPscores)werethefollow- ing:logaqueoussolubility(logS),logPandPPB.Thesedescriptors hadVIPscoresabove1.3.Theresultconfirmsthethermodynamic
Table2
SummaryoftheperformanceparametersforthefinalQSPRmodels*.
Model Permeability Membraneretention Permeability(in logsale)
RMSEC 2.87 4.37 0.04
RMSECV 5.06 8.14 0.08
RMSEP 5.15 7.05 0.11
R2 0.95 0.97 0.95
Q2CV 0.87 0.91 0.86
R2P 0.86 0.94 0.85
Q2F1 0.86 0.96 0.75
Q2F2 0.86 0.93 0.75
Q2F3 0.85 0.93 0.79
CCCTrain 0.98 0.99 0.97
CCCCV 0.93 0.95 0.93
CCCExt 0.92 0.97 0.89
MAETrain 2.15 3.28 0.04
MAECV 3.79 4.90 0.06
MAEExt 3.92 5.74 0.08
r2m 0.80 0.89 0.58
r¯2m 0.76 0.88 0.65
r2m 0.07 0.01 0.13
r02−r02 0.02 0.00 0.07k 0.87 0.96 1.09
k’ 0.91 0.99 1.01
Y-SCRCV 0.02 0.02 –
Y-SCRP. 0.14 0.04 –
PLSCOMP. 5 8 –
*RMSEC–rootmeansquarederrorofcalibration,RMSECV–rootmeansquared errorofcross-validation,RMSEP–rootmeansquarederrorofexternalvalidation, R2-goodnessoffitforthecalibration,Q2CV-goodnessoffitforthecross-validation, R2P-goodnessoffitfortheexternalvalidation,QF12–F1typegoodnessoffitforthe externalvalidation,QF22–F2typegoodnessoffitfortheexternalvalidation,QF32– F3typegoodnessoffitfortheexternalvalidation,CCC(Train/CV/Ext)–coefficient ofconcordanceforthetraining/CV/externaltestset,MAE(Train/CV/Ext)–mean absoluteerrorforthetraining/CV/externaltestset,r2mandr2m–basedonthe workofRoyetal.[52],
r02−r02istheabsolutedifferenceofther20valueswithout interceptinthecaseofobservedvs.predictedandpredictedvs.observedvalues, kandk’-theslopeoftheregressionlines(withorwithoutpassingthroughthe origin),Y-SCRCV-goodnessoffitforthecrossvalidationusingYscrambling,Y-SCR P-goodnessoffitforthetestpredictionusingY-scrambling,PLSCOMP.–numberof PLScomponentsinthemodel.Fig.5.Predictionofthemembraneretention.Predictedvaluesareplottedagainst theexperimentalmembraneretentionvalues.Thetestsamplesaremarkedwith reddiamondsandthecalibrationonesaremarkedwithgreycircles.Thecalibration curveismarkedwiththeredline.
behavioroftheMRparameterforthetestedcompoundsinthatit relatestotheirdistributionbetweenthedonor,acceptorandlipid membrane.TheprimaryrelevanceofthelogPandlogSparame- terssuggeststhedominanceoftheneutralformofthecompounds, whichalsosupportsthespecificityoftheinsilicodescription,as theprotondissociationofdrugmoleculesinthemembraneistypi-
callydepressed.ThefactthatlogPismorefavorablethanlogDpH7.4 asa descriptor for membrane retention confirms theresultsof ourpreliminaryanalysis(SeeFigureB1.,AppendixB).Theweighted appearanceofPPBcanbeexplainedbythelipophilicity-dependent natureofthefreeformofthedrug,whichisconsistentwiththe generalkineticdescriptionofdrugtransport.Additionally,linear regressionanalysiswascarried outin thecaseof logS andPPB againstMRandonlyweakcorrelationswerefoundwiththesquared correlationcoefficientsof0.232and0.189respectivelywhichalso verifiestheneedformorecomplexmodelsthanthelinearregres- sionofonephysicochemicalparameteratatime(SeeFigureB3., AppendixBandTableA2.,AppendixA).
Y-scramblingwasalsousedtocheckthepossibilityofover- fitting.Themodelperformancesstronglydecreasedwiththeuse ofrandomizedYvalues.TheQ2CVwas0.02,whiletheQ2fortest predictionwas0.04.Thepermutationtest(with50iterations)has foundasignificantdifferencebetweenourfinalmodelandtheran- domizedone(at␣=0.05level),verifyingourmodel.
Thedetailedsummaryoftheperformanceparametersofthe finalmodels(permeabilityandmembraneretention)isshownin Table2.Allparametersaregoodandfulfillthesuggestedconditions fortheacceptanceofthemodel:R2 >0.7,QFn2>0.6,CCC>0.85, r2m >0.5,r2m< 0.2[42,51],and
r02−r02 <0.2.Based onthe performancesofthemodels,wearerecommendingtheuseofper- meabilityvaluesinlinearscaleforfurthercalculations.4. Conclusion
Twoquantitativestructure-propertyrelationshipmodelswere developedfortheinvestigationofcornealpermeabilityandmem- braneretention.TheQSPRmodelswerebasedonexperimentally determinedcorneal-PAMPAvalues.Partialleastsquaresregression wasappliedfor theprocess. Thefinal models containedclassi- calmoleculardescriptorsandECFPfingerprintsin combination.
Themodelsincluded143and180APIsforPeandMRprediction, respectively.Acomplexvalidationprotocol(includinginternaland external phases)was carried outto provide robustand appro- priatepredictionsforthepermeabilityand membraneretention values.Thegoodnessofthemodels(R2)wereabove0.90inboth cases and theywere not lower than0.85 for validation either.
Thepermutationtests showedthatourmodelsare notoverfit- tedandtheyareclearlyapplicabletoprovidegoodpredictionsfor thecornealpermeabilityoftheAPIs.Thisis,toourknowledge,the firstsuchinsilicomodelreportedsofar.Theresultsalsoshowed thattherewasnosignificantcorrelationbetweentheexperimental corneal-PAMPA values and the existing classical in silicophar- macokineticallyrelevantdrugtransport models,suchasCaco-2 permeability,jejunalpermeability andtheblood-brainpartition coefficient(logBB).Thus,ourmodelscanbegoodcandidatestopro- videthecornealpermeabilityandcornealmembraneretentionof compoundsinaveryshorttimewithappropriateprecision.
Authors’contributions
The manuscript was written through contributions of all authors.Allauthorshavegivenapprovaltothefinalversionofthe manuscript.
DeclarationofCompetingInterest
Theauthorsreportnodeclarationsofinterest.